Dilution Formula: Dilution is the process of reducing the pH of a chemical substance, which can exist in the form of a gas, vapor, or solution. This process entails lowering the concentration of a solute within a solution by typically mixing it with a solvent. To perform dilution, additional solvent is introduced without the addition of more solute, and thorough mixing is essential to ensure uniform distribution throughout the resulting solution.
As an illustration, if you initially have 10 grams of salt dissolved in 1 liter of water, you have a specific salt concentration. When you add an additional 1 liter of water to this solution, the salt concentration decreases, but the total amount of salt remains at 10 grams.
To create a specific volume of a dilute solution, you can use the dilution formula:
C1V1 = C2V2
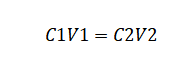
Where:
V1 represents the volume of the stock solution required to prepare the new solution.
V2 is the final volume of the solution.
C1 denotes the concentration of the stock solution.
C2 is the final concentration of the stock solution.
Also Check - Theoretical Yield Formula
Dilution Formula Solved Examples
Example 1:
A chemist needs a 1.5M hydrochloric acid solution for a chemical reaction, but the available solution is 6M HCl. What volume of the 6M HCl solution must be diluted to yield 5 liters of a 1.5M HCl solution?
Solution:
Initial HCl concentration (C1) = 6M
Final HCl concentration (C2) = 1.5M
Final volume of the solution (V2) = 5 liters
We need to determine the initial volume (V1) required.
Substituting the values into the formula:
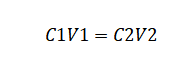
V1 = (C2 * V2) / C1
V1 = (1.5 * 5.0) / 6
V1 = 1.25 liters
Therefore, 1.25 liters of the 6M HCl solution must be diluted to obtain 5 liters of a 1.5M HCl solution.
Also Check - Degree Of Unsaturation Formula
Example 2: How much of a 12M sulfuric acid solution do you need to prepare a 500 milliliter (0.5-liter) solution of 2M sulfuric acid?
Solution:
Initial sulfuric acid concentration (C1) = 12M
Final sulfuric acid concentration (C2) = 2M
Final volume of the solution (V2) = 0.5 liters (500 milliliters)
We need to calculate the initial volume (V1) required.
Using the dilution formula:
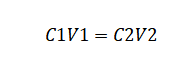
V1 = (C2 * V2) / C1
V1 = (2 * 0.5) / 12
V1 = 0.0833 liters (or 83.3 milliliters)
So, you would need to mix 83.3 milliliters of the 12M sulfuric acid solution with additional water to prepare 500 milliliters of a 2M sulfuric acid solution.
Also Check - Vapor Pressure Formula
Example 3: How much of the concentrated bleach solution containing 10% sodium hypochlorite (NaClO) should be diluted to create a 500 milliliter solution with a 2% sodium hypochlorite concentration?
Solution:
Initial sodium hypochlorite concentration (C1) = 10%
Final sodium hypochlorite concentration (C2) = 2%
Final volume of the solution (V2) = 500 milliliters (0.5 liters)
We want to find the initial volume (V1) required:
Using the dilution formula:
V1 = (C2 * V2) / C1
V1 = (2% * 0.5 L) / 10%
V1 = 0.01 L (or 10 milliliters)
To prepare 500 milliliters of a 2% sodium hypochlorite solution, you should dilute 10 milliliters of the 10% concentrated bleach solution with additional water.
Also Check - Gibbs Free Energy Formula
Example 4: How much of the 8.0M hydrofluoric acid (HF) stock solution should be used, and how much water should be added to prepare 250 milliliters of a 1.0M HF solution for the laboratory experiment?
Solution:
Initial hydrofluoric acid concentration (C1) = 8.0M
Final hydrofluoric acid concentration (C2) = 1.0M
Final volume of the solution (V2) = 250 milliliters (0.25 liters)
We need to calculate the initial volume (V1) required:
Using the dilution formula:
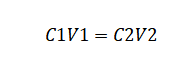
V1 = (C2 * V2) / C1
V1 = (1.0M * 0.25 L) / 8.0M
V1 = 0.03125 L (or 31.25 milliliters)
So, you should measure 31.25 milliliters of the 8.0M hydrofluoric acid solution and then add enough water to reach a total volume of 250 milliliters. This will result in a 1.0M HF solution for your experiment.
These examples demonstrate how to use the dilution formula to prepare solutions of different concentrations by mixing concentrated solutions with the appropriate amount of solvent. It's a fundamental concept in chemistry and is widely used in laboratory work and various industries.
Dilution Formula FAQs
What is the dilution formula?
Why is dilution important in chemistry?
How does the dilution formula work?
What happens to the total amount of solute during dilution?













