Internal Energy Formula: Internal energy is also known as thermal energy. It represents the energy of a substance resulting from the kinetic and potential energies associated with the random motion of all the particles that compose the substance. The energy encompassed within a system is referred to as internal energy and is denoted by the symbol 'U.'
In essence, internal energy pertains to the energy linked with the chaotic motion or disordered arrangement of particles within the system, and it is typically measured in kilojoules (KJ) or joules (J). We can calculate internal energy using the following formula.
Also Check - Molecular Speed Formula
The change in internal energy is equivalent to the difference between the heat transferred in a system and the work done on or by the system (PV). The formula for calculating this change in internal energy is a crucial tool in thermodynamics.
On this page, we will discuss the various aspects related to internal energy, including the internal energy formula, ideal gas, specific internal energy formula, change in internal energy equation, and the total internal energy formula.
Also Check - Mole Fraction Formula
Internal Energy Equation
In accordance with the first law of thermodynamics, the total energy of the universe remains unchanged.
Furthermore, the change in the internal energy of a system is equal to the sum of the heat transferred and the work done. Additionally, the heat added or removed from the system equals the combination of the change in the internal energy and the PV work done.
The formula for the internal energy of a gas is expressed as:
Q = ΔU + W … (1)
This equation can also be rearranged to derive the formula for the change in internal energy:
ΔU = Q - PV … (2)
Equation (2) is commonly referred to as the total internal energy formula or the change in internal energy equation.
Also Check - Ionization Energy FormulaTotal Internal Energy Formula
The total internal energy formula encompasses the cumulative energies, including translational, rotational, binding, and chemical energies, of all the particles within the system.
Internal energy also incorporates potential energy, which is stored in the form of chemical bonds, attractions, or repulsions. Additionally, it includes kinetic energy originating from the motion, translation, rotation, and vibration of particles within the system.
Change in Internal Energy Formula
The formula for the change in internal energy is as follows:
ΔU = Q + W
Here:
U = the total change in internal energy within the system
Q = the heat exchanged between the system and its surroundings (external to the system)
W = work done on or by the system
Internal Energy Formula for Ideal Gas
In thermodynamics, although the change in internal energy (ΔU) is often emphasized for ideal gases, the absolute internal energy (U) can also be determined.
To calculate the absolute internal energy, consider a gas enclosed in a constant-volume chamber that is cooled to absolute zero. In this scenario, all particles are stationary, and the gas possesses no internal energy initially. However, heat (Q) is added at a constant volume until the gas reaches a temperature 'T.'
All the heat needed to raise the gas's temperature to 'T' ultimately contributes to the internal energy (U). Therefore, at temperature 'T,' the gas's internal energy (U) is given by:
U = cv * T —----- (3)
Here:
cv = the specific isochoric heat capacity, which may be temperature-dependent for ideal gases.
Formula for Change in Internal Energy
To derive the formula for the internal energy of a gas (equation 4), we start with equation 3 and express it in terms of the change in internal energy:
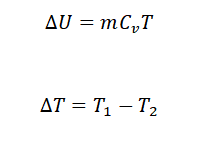
Here, equation (4) represents the specific internal energy formula.
Analogy Between Internal Energy and Gravitational Potential Energy
The internal energy of ideal gases can be likened to the gravitational potential energy of an object. Just as gravitational potential energy represents the potential energy state of an object at a certain height 'h,' internal energy signifies the kinetic energy state of an ideal gas at a given temperature 'T.'
Specific potential energy for a given height 'h' is determined by the object's mass 'm,' analogous to specific internal energy being determined by the gas's mass 'm' at a given temperature 'T.'
The specific relationship between potential energy and height 'h' is defined by gravitational acceleration 'g' (which varies with location).
The relationship between internal energy 'U' and temperature 'T' is determined by the specific heat capacity at constant volume, denoted as 'cv,' which varies according to the gas's properties.
Internal Energy Formula Solved Example
Example 1: Determining Internal Energy Change in an Internal Combustion Engine
Problem: Calculate the change in the internal energy of the working fluid in an internal combustion engine, and specify whether it is a gain or loss. Given that during the compression stroke, 80 KJ/kg of heat is rejected to the cooling water, and the work input is 200 KJ/kg.
Solution:
Heat rejected to the cooling water: Q = -80 KJ/kg
Work input: W = -200 KJ/kg
Using the formula:
Q = ΔU - W
-80 = ΔU - 200
ΔU = -80 + 200
ΔU = 120 KJ/kg
Hence, the internal energy increases by ΔU = 120 KJ/kg.
Internal Energy Formula FAQs
What is internal energy?
How is internal energy measured?
What is the formula for internal energy change?
What does ΔU represent in the internal energy formula?













