Coordination Compound NEET Notes: Coordination compounds are a vital topic in inorganic chemistry, especially for NEET aspirants with the exam scheduled on 3 May 2026. These compounds consist of a central metal atom or ion bonded to surrounding molecules or ions called ligands through coordinate bonds.
Coordination Compound NEET Notes Overview
Coordination compounds are formed when a metal ion binds with ligands through coordinate bonds. These compounds have unique properties and applications in various fields like biology, medicine, and industrial chemistry. For NEET aspirants, this topic is important as questions are frequently asked in the exam.
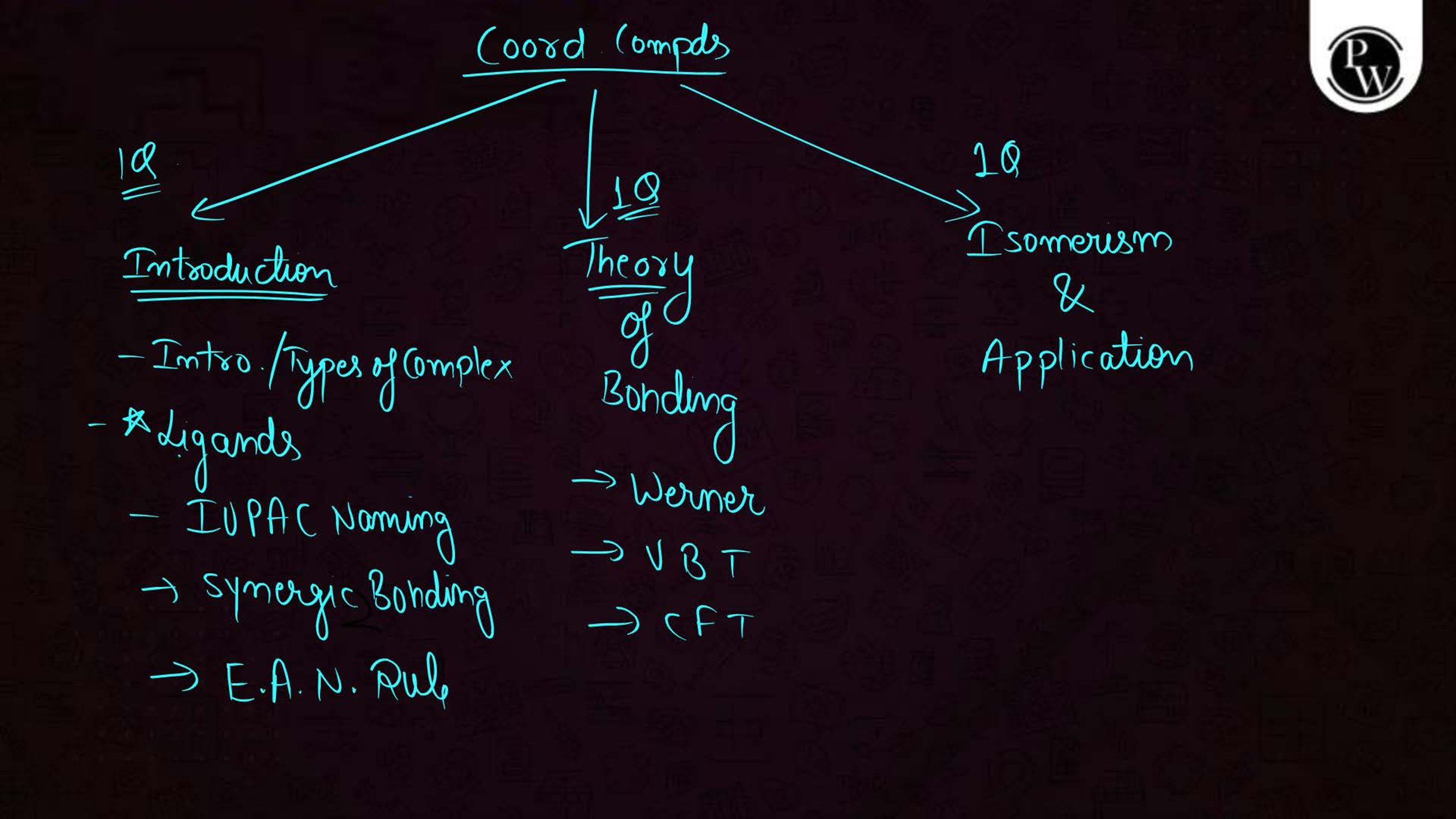
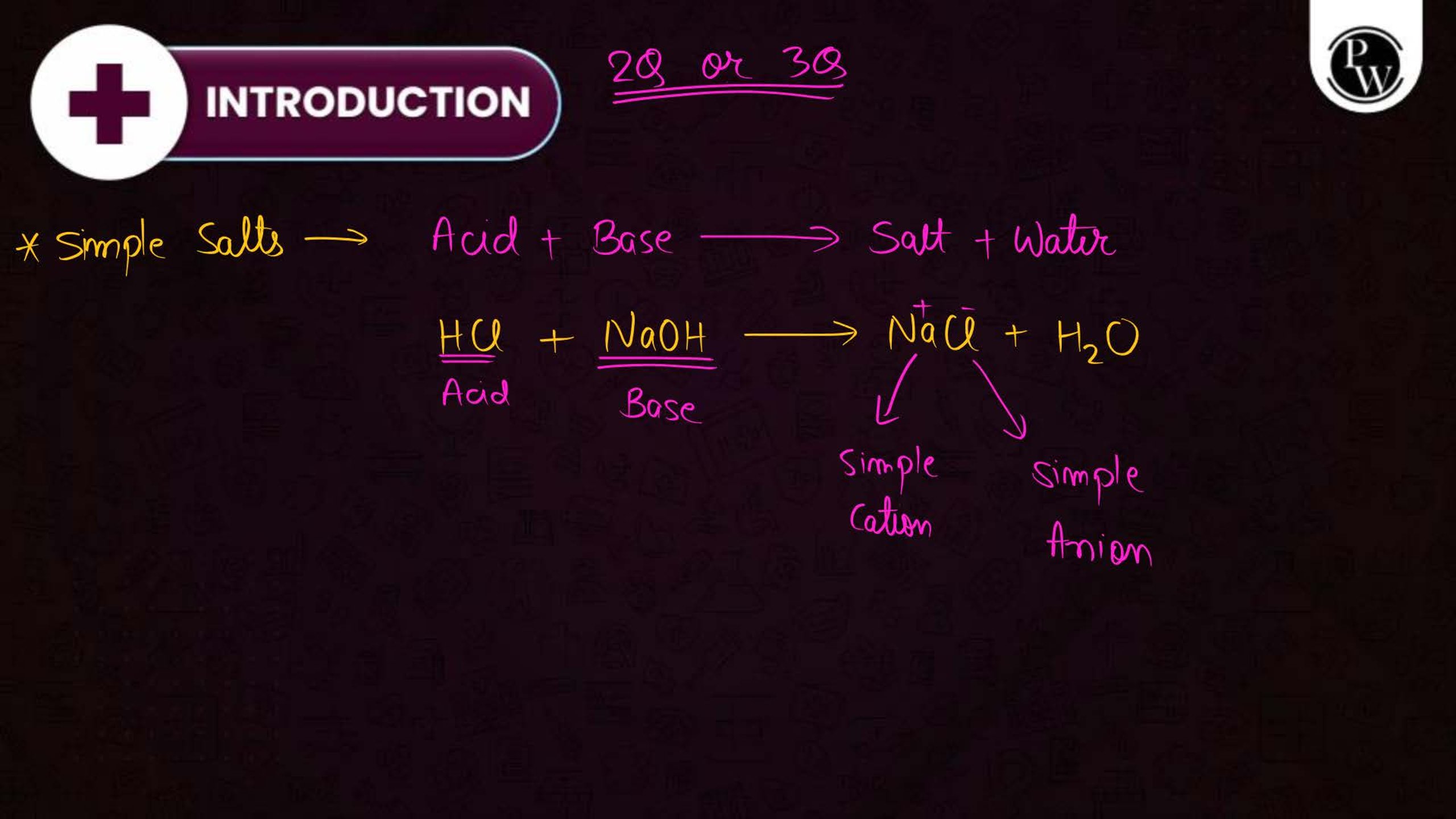
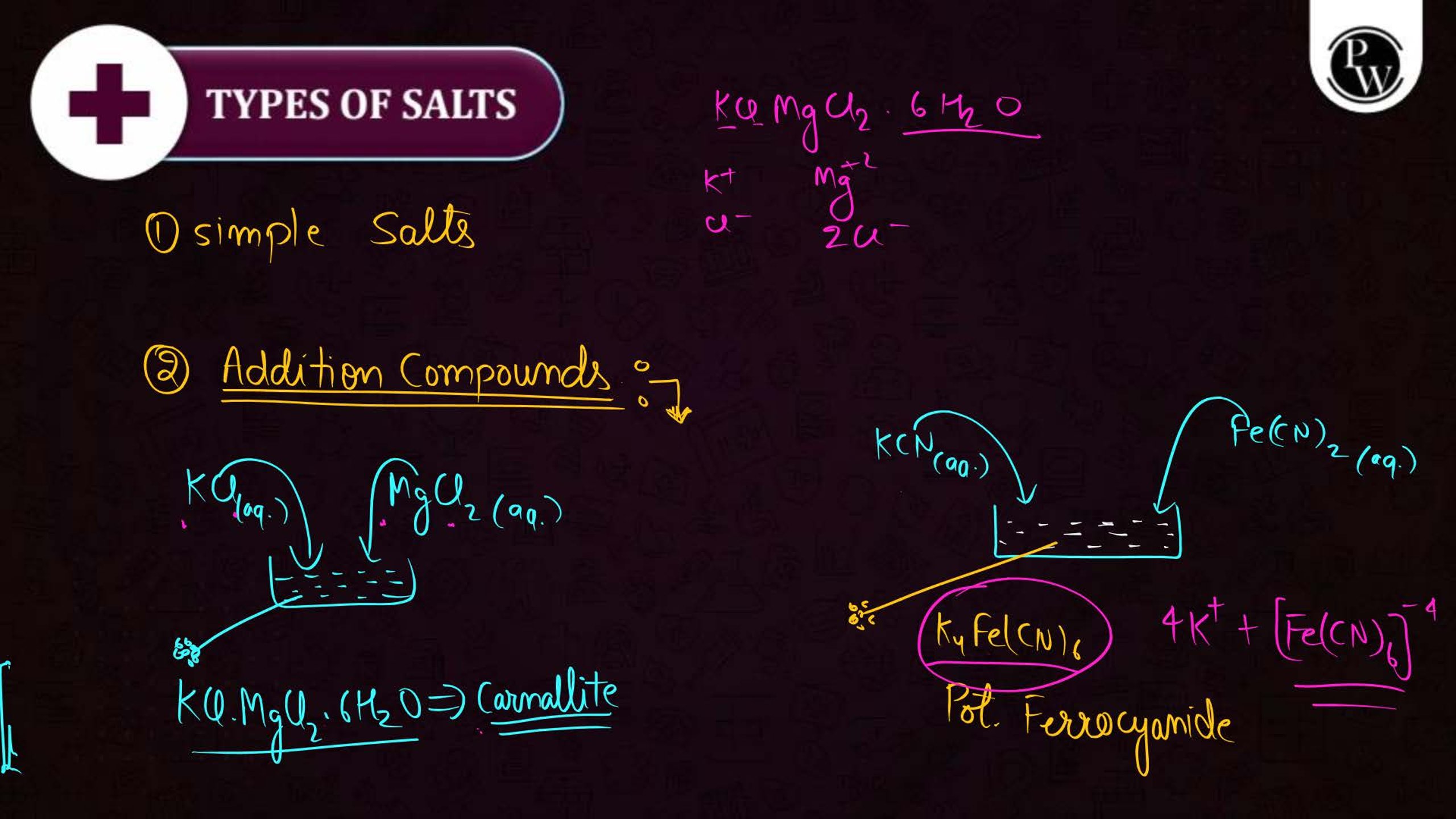

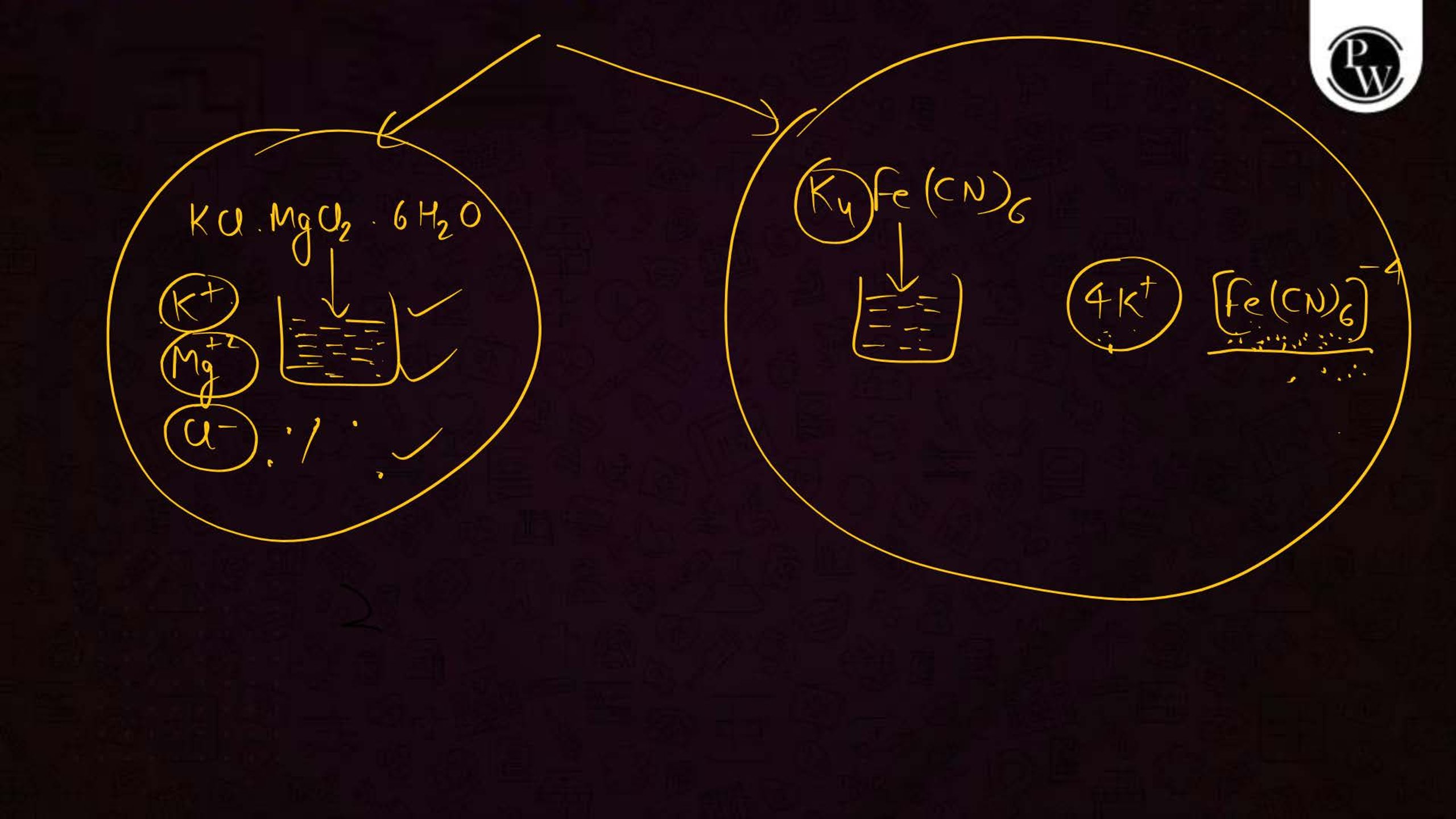




What is the Coordination Compound?
A coordination compound consists of a central metal atom or ion surrounded by a group of molecules or ions called ligands. These ligands are attached to the central atom through coordinate bonds. Coordination compounds are commonly used in medicines, industries, and chemical analysis.
Weightage of Coordination Compound NEET Notes - Last 5 Years
The table below summarizes the importance of this chapter in NEET based on the last five years (2020–2024):
| Weightage Following Previous Year’s Trends (2020-2024) | ||||||
| Chapter | Easy | Medium | Hard | Total | Average Q/yr | Weightage |
| Coordination Compounds | 2 | 5 | 4 | 11 | 2.20 | 4.18% |
| NEET Chapter Wise Weightage 2025 for Chemistry | |
| Topics | Weightage (%) |
| Coordination Compounds | 5% |
Importance of Coordination Compound NEET Notes
This chapter is important because it focuses on basic concepts that can help you understand other topics in chemistry. Questions from this chapter are direct and scoring.
- It simplifies complex reactions involving metal ions.
- It is widely used in real-world applications like catalysis, medicines, and color pigments.
- Scoring well in this chapter boosts your overall chemistry score.
Tips for Coordination Compound NEET Notes
Studying coordination compounds effectively can save you time and effort. Below are some tips to help you master this topic:
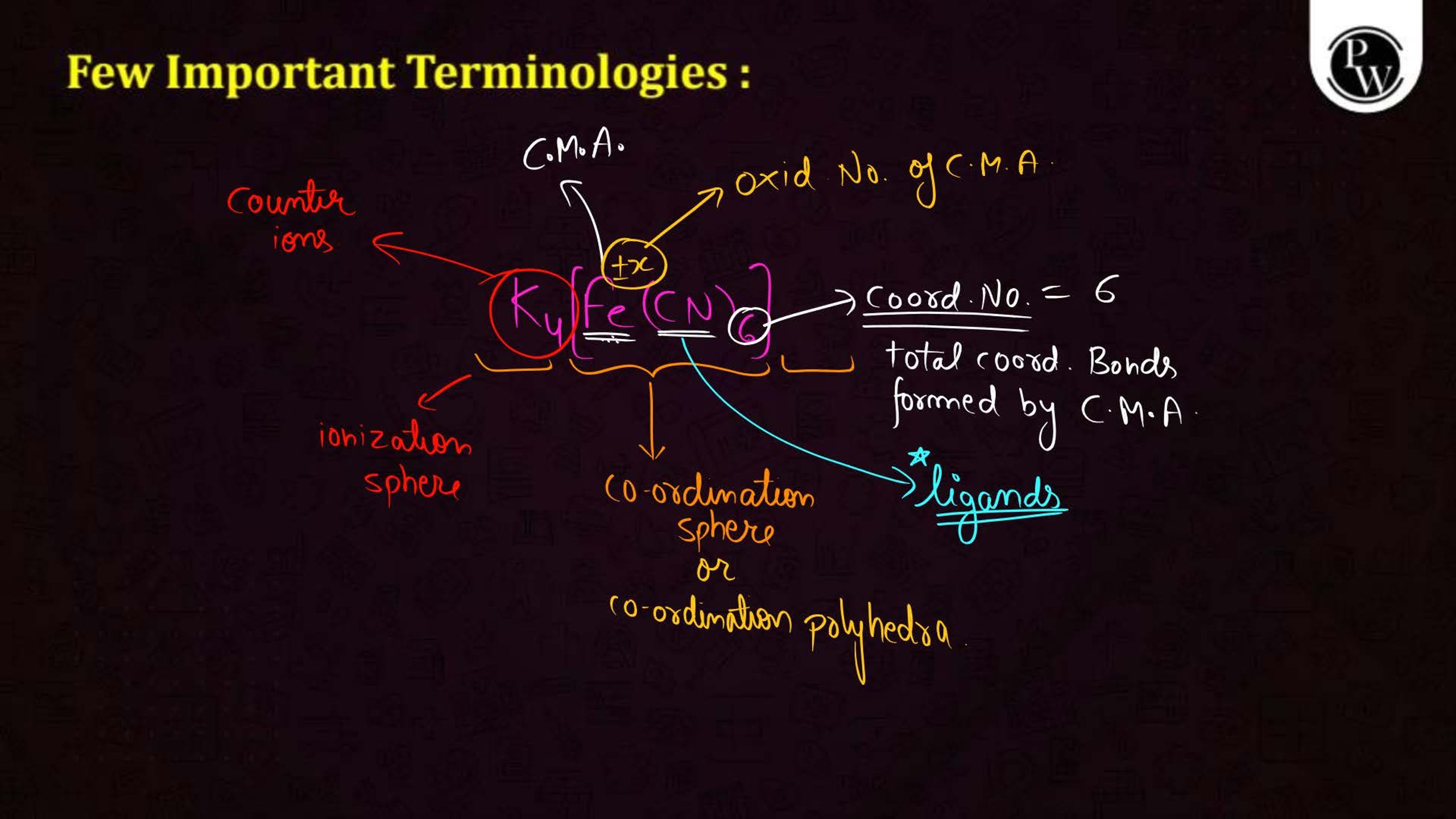
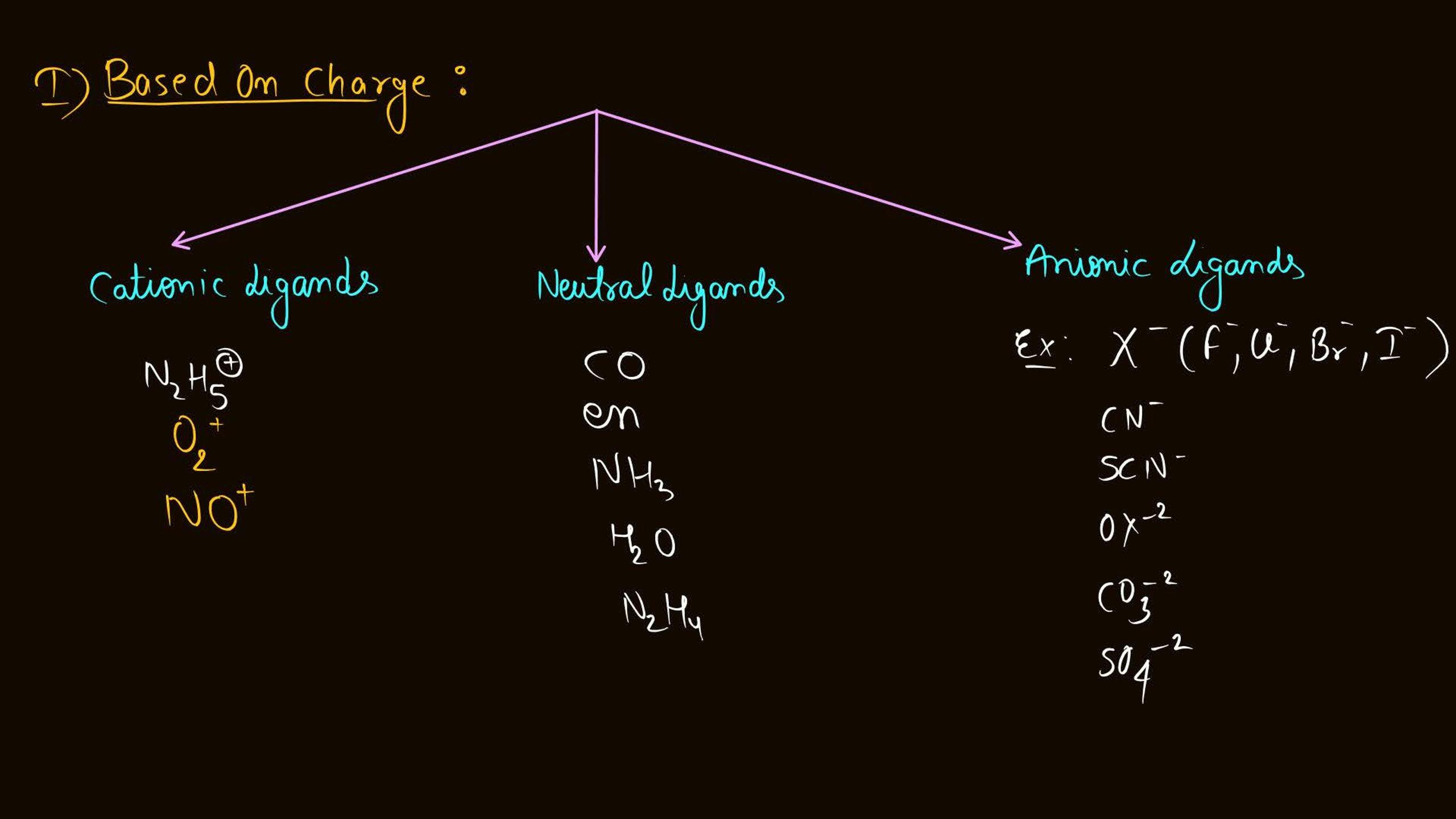
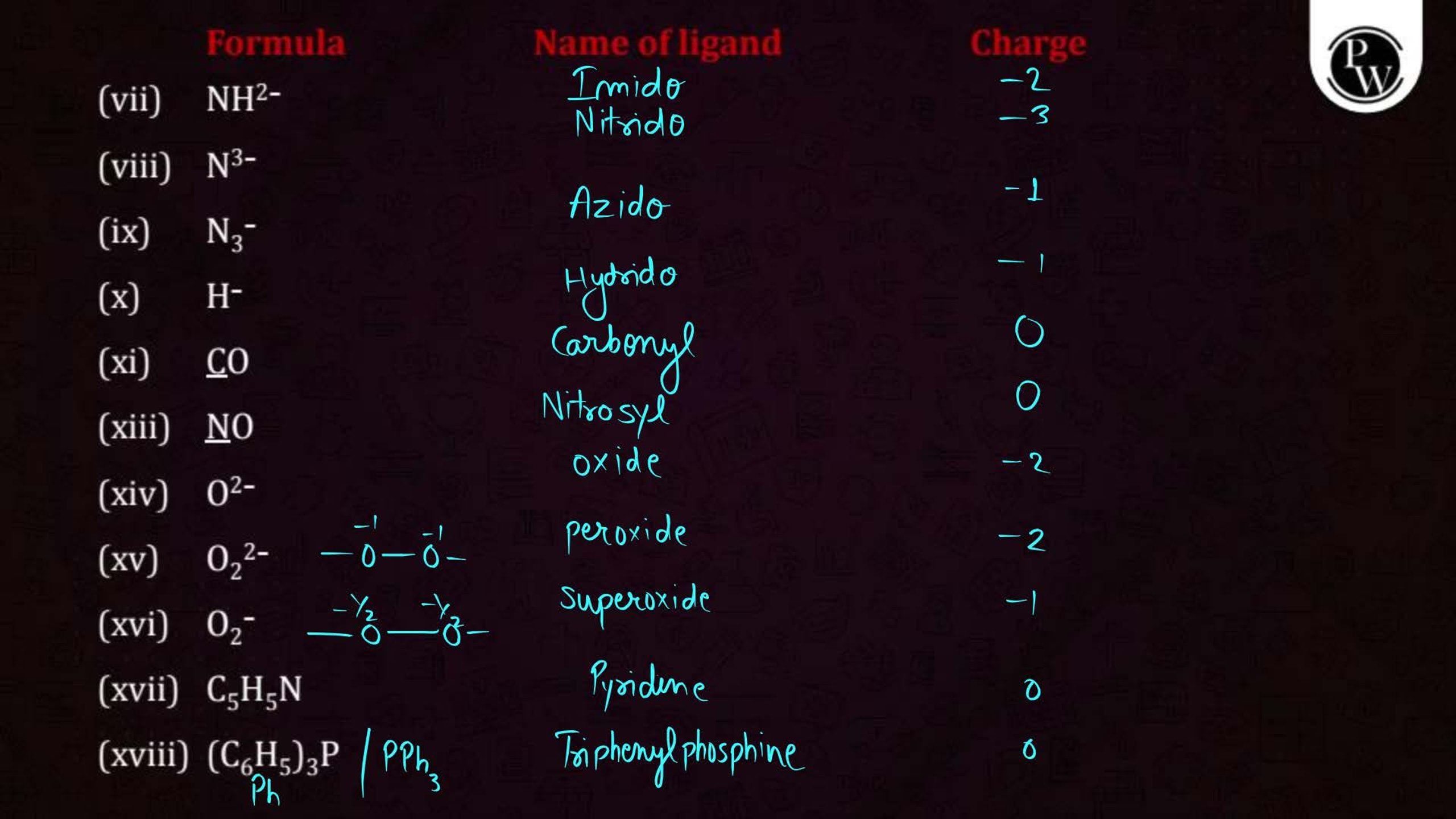
- Learn definitions and terminology: Focus on terms like ligands, coordination number, and chelation.
- Understand IUPAC naming Rules : Practice naming coordination compounds systematically.
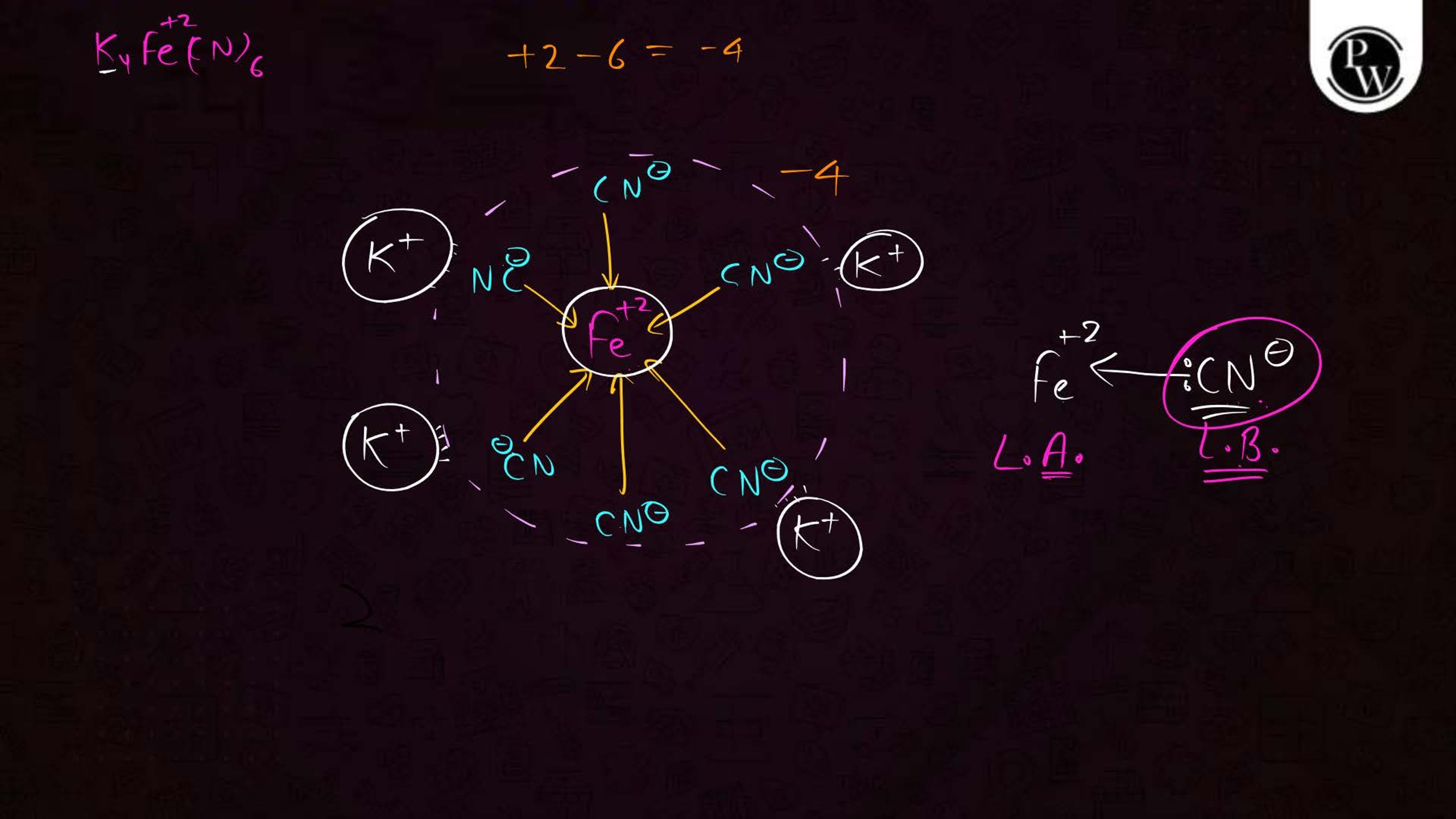
- Memorize common examples: Know compounds like [Fe(CN)6]3– and [Co(NH3)6]3+.
- Focus on isomerism : Isomers are commonly asked in NEET.
- Revise regularly: Short notes and flashcards can help with quick revisions.
Prepare for NEET with PW Online NEET Coaching ! Get comprehensive guidance with structured lessons, in-depth concepts, and interactive classes designed to help you succeed.



