
NCERT Solutions for Class 12 Chemistry Chapter 7 The P Block Elements contains all the questions with detailed and step-by-step solutions. Students are advised to carefully read the complete theory of Chapter 7 before solving the NCERT questions to build a strong conceptual understanding.
This chapter is very important for the board exam, and practicing these solutions will help you revise key concepts, reactions, trends, and properties effectively. Since the exam is scheduled on 28 February 2026, preparing this chapter thoroughly will boost your confidence and help you perform well in the exam.
NCERT Solutions for Class 12 Chemistry Chapter 7
Answer the following Questions of NCERT Solutions for class-12 Chemistry Chapter 7 P Block Elements
Question 1. Why are pentahalides more covalent than trihalides? Solution : In pentahalides, the oxidation state is +5 and in trihalides, the oxidation state is +3. Since the metal ion with a high charge has more polarizing power, pentahalides are more covalent than trihalides.NCERT Solutions For Class 12 Chemistry Chapter 2
Question 2. Why is BiH3 the strongest reducing agent among all the hydrides of Group 15 elements? Solution : As we move down a group, the atomic size increases and the stability of the hydrides of group 15 elements decreases. Since the stability of hydrides decreases on moving from NH3 to BiH3, the reducing character of the hydrides increases on moving from NH3 to BiH3.NCERT Solutions For Class 12 Chemistry Chapter 3
Question 3. Why is N2 less reactive at room temperature? Solution : The two N atoms in N2 are bonded to each other by very strong triple covalent bonds. The bond dissociation energy of this bond is very high. As a result, N2 is less reactive at room temperature. Question 4. Mention the conditions required to maximise the yield of ammonia. Solution : Ammonia is prepared using the Haber’s process. The yield of ammonia can be maximized under the following conditions: (i) High pressure (∼ 200 atm) (ii) A temperature of ∼700 K (iii) Use of a catalyst such as iron oxide mixed with small amounts of K2O and Al2O3NCERT Solutions For Class 12 Chemistry Chapter 4
Question 5. How does ammonia react with a solution of Cu2+? Solution : NH3 acts as a Lewis base. It donates its electron pair and forms a linkage with a metal ion. Question 6. What is the covalence of nitrogen in N2O5? Solution :
Question 6. What is the covalence of nitrogen in N2O5? Solution :  From the structure of N2O5, it is evident that the covalence of nitrogen is 4.
From the structure of N2O5, it is evident that the covalence of nitrogen is 4.
NCERT Solutions For Class 12 Chemistry Chapter 5
Question 7. The bond angle in PH + 4 is higher than that in PH3. Why? Solution : In PH3, P is sp3 hybridized. Three orbitals are involved in bonding with three hydrogen atoms and the fourth one contains a lone pair. As lone pair-bond pair repulsion is stronger than bond pair-bond pair repulsion, the tetrahedral shape associated with sp3 bonding is changed to pyramidal. PH3 combines with a proton to form PH + 4 in which the lone pair is absent. Due to the absence of a lone pair in PH + 4 , there is no lone pair-bond pair repulsion. Hence, the bond angle in PH + 4 is higher than the bond angle in PH3. Question 8. What happens when white phosphorus is heated with concentrated NaOH solution in an inert atmosphere of CO2? Solution : White phosphorous dissolves in boiling NaOH solution (in a CO2 atmosphere) to give phosphine, PH3.
Question 8. What happens when white phosphorus is heated with concentrated NaOH solution in an inert atmosphere of CO2? Solution : White phosphorous dissolves in boiling NaOH solution (in a CO2 atmosphere) to give phosphine, PH3. 

NCERT Solutions For Class 12 Chemistry Chapter 6
Question 10. Write a balanced equation for the hydrolytic reaction of PCl5 in heavy water. Solution :
 Since there are three OH groups present in H3PO4, its basicity is three i.e., it is a tribasic acid.
Since there are three OH groups present in H3PO4, its basicity is three i.e., it is a tribasic acid.

 (ii)
(ii) 
 Therefore, ozone acts as a powerful oxidising agent.
Therefore, ozone acts as a powerful oxidising agent.

 Both S−O bonds are equal in length (143 pm) and have a multiple bond character.
Both S−O bonds are equal in length (143 pm) and have a multiple bond character.

- It is used in fertilizer industry. It is used to make various fertilizers such as ammonium sulphate and calcium super phosphate.
- It is used in the manufacture of pigments, paints, and detergents.
- It is used in the manufacture of storage batteries.
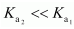 for H2SO4 in water? Solution :
for H2SO4 in water? Solution :  It can be noticed that
It can be noticed that  This is because a neutral H2SO4 has a much higher tendency to lose a proton than the negatively charged
This is because a neutral H2SO4 has a much higher tendency to lose a proton than the negatively charged  . Thus, the former is a much stronger acid than the latter.
. Thus, the former is a much stronger acid than the latter. Coloured substances + [O] → Oxidized colourless substance
Coloured substances + [O] → Oxidized colourless substance NO and HNO3 are produced in small amounts. These are impurities that can be removed on passing nitrogen gas through aqueous sulphuric acid, containing potassium dichromate.
NO and HNO3 are produced in small amounts. These are impurities that can be removed on passing nitrogen gas through aqueous sulphuric acid, containing potassium dichromate.

 (2)
(2) 
| White phosphorus | Red Phosphorus |
| It is a soft and waxy solid. It possesses a garlic smell. | It is a hard and crystalline solid, without any smell. |
| It is poisonous. | It is non-poisonous. |
| It is insoluble in water but soluble in carbon disulphide. | It is insoluble in both water and carbon disulphide. |
| It undergoes spontaneous combustion in air. | It is relatively less reactive. |
In both solid and vapour states, it exists as a P4 molecule.  |
It exists as a chain of tetrahedral P4 units.  |

 Step (iii): SO3 produced is absorbed on H2SO4 to give H2S2O7 (oleum).
Step (iii): SO3 produced is absorbed on H2SO4 to give H2S2O7 (oleum).  This oleum is then diluted to obtain H2SO4 of the desired concentration. In practice, the plant is operated at 2 bar (pressure) and 720 K (temperature). The sulphuric acid thus obtained is 96-98% pure.
This oleum is then diluted to obtain H2SO4 of the desired concentration. In practice, the plant is operated at 2 bar (pressure) and 720 K (temperature). The sulphuric acid thus obtained is 96-98% pure.- It combines with water vapour present in the atmosphere to form sulphuric acid. This causes acid rain. Acid rain damages soil, plants, and buildings, especially those made of marble.
- Even in very low concentrations, SO2 causes irritation in the respiratory tract. It causes throat and eye irritation and can also affect the larynx to cause breathlessness.
- It is extremely harmful to plants. Plants exposed to sulphur dioxide for a long time lose colour from their leaves. This condition is known as chlorosis. This happens because the formation of chlorophyll is affected by the presence of sulphur dioxide.
- It is used for purifying water.
- It is used as a bleaching agent.
 (ii)
(ii)  Question 64. How can you prepare Cl2 from HCl and HCl from Cl2? Write reactions only. Solution : (i) Cl2 can be prepared from HCl by Deacon’s process.
Question 64. How can you prepare Cl2 from HCl and HCl from Cl2? Write reactions only. Solution : (i) Cl2 can be prepared from HCl by Deacon’s process. 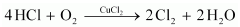 (ii) HCl can be prepared from Cl2 on treating it with water.
(ii) HCl can be prepared from Cl2 on treating it with water. 
 . Later, he realized that the first ionization energy of oxygen (1175 kJ/mol) and Xe (1170 kJ/mol) is almost the same. Thus, he tried to prepare a compound with Xe and PtF6. He was successful and a red-coloured compound,
. Later, he realized that the first ionization energy of oxygen (1175 kJ/mol) and Xe (1170 kJ/mol) is almost the same. Thus, he tried to prepare a compound with Xe and PtF6. He was successful and a red-coloured compound,  was formed.
was formed. (ii) PCl3
(ii) PCl3  (iii) Ca3P2
(iii) Ca3P2  (iv) Na3PO4
(iv) Na3PO4  (v) POF3
(v) POF3 
 (ii)
(ii) 

 (ii) XeOF4 can be prepared using XeF6.
(ii) XeOF4 can be prepared using XeF6. 
 Solution : (i) XeF4 is isoelectronic with
Solution : (i) XeF4 is isoelectronic with  and has square planar geometry.
and has square planar geometry.  (ii) XeF2 is isoelectronic to
(ii) XeF2 is isoelectronic to  and has a linear structure.
and has a linear structure.  (iii) XeO3 is isostructural to
(iii) XeO3 is isostructural to  and has a pyramidal molecular structure.
and has a pyramidal molecular structure.  Question 74. Why do noble gases have comparatively large atomic sizes? Solution : Noble gases do not form molecules. In case of noble gases, the atomic radii corresponds to van der Waal’s radii. On the other hand, the atomic radii of other elements correspond to their covalent radii. By definition, van der Waal’s radii are larger than covalent radii. It is for this reason that noble gases are very large in size as compared to other atoms belonging to the same period. Question 75. List the uses of Neon and argon gases. Solution : Uses of neon gas:
Question 74. Why do noble gases have comparatively large atomic sizes? Solution : Noble gases do not form molecules. In case of noble gases, the atomic radii corresponds to van der Waal’s radii. On the other hand, the atomic radii of other elements correspond to their covalent radii. By definition, van der Waal’s radii are larger than covalent radii. It is for this reason that noble gases are very large in size as compared to other atoms belonging to the same period. Question 75. List the uses of Neon and argon gases. Solution : Uses of neon gas:
- It is mixed with helium to protect electrical equipments from high voltage.
- It is filled in discharge tubes with characteristic colours.
- It is used in beacon lights.
- Argon along with nitrogen is used in gas-filled electric lamps. This is because Ar is more inert than N.
- It is usually used to provide an inert temperature in a high metallurgical process.
- It is also used in laboratories to handle air-sensitive substances.