
Uric Acid Formula: Carbon is a nonmetallic element with a tetravalent nature, serving a significant purpose for humanity. Its name, "carbon," finds its roots in the Latin word "carbo," meaning charcoal or ember. Despite its minimal presence, comprising only 0.025% of the Earth's crust and 0.03% in the atmosphere, carbon plays a vital role in human existence. Its abundance and capacity to form polymers at room temperature make it a fundamental component in all living organisms. Limestones and dolomite serve as primary sources of inorganic carbon.
On the other hand, Nitrogen, another nonmetal, dominates the Earth's atmosphere, constituting up to 78% of its composition, rendering it the most abundant element. It exists within all living beings in the form of nucleic acids, specifically DNA and RNA. Nitrogen, denoted by its atomic number 7 and symbol N, has a ubiquitous presence in various pharmaceutical drugs. The credit for its discovery goes to the Scottish physician Daniel Rutherford, who identified it in 1772.
Uric Acid
Uric acid is a chemical compound produced during the breakdown of purines, which naturally occur in the body and can also be found in certain foods. These purine-rich materials are particularly abundant in substances such as alcohol, shellfish, and liver. Uric acid is generated as a byproduct when DNA is broken down, representing one of the waste products created within the body. Elimination of uric acid from the body takes place through either the process of urination or via bowel movements. Elevated levels of uric acid in the bloodstream often indicate an unhealthy lifestyle or malfunctioning kidneys.
Swedish chemist Carl Wilhelm Scheele is credited with the discovery of uric acid in 1776 when he successfully isolated it from kidney stones. Despite its initial discovery, uric acid was not synthesized until 1882, when Ukrainian chemist Ivan Horbaczewski achieved this by melting urea and glycine.
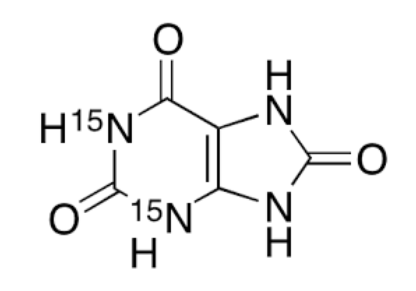
Uric Acid Structure
Uric acid comprises a combination of Carbon, Hydrogen, Nitrogen, and Oxygen elements, with a chemical formula of C 5 H 4 N 4 O 3 .Its formation occurs as a result of the breakdown of purines. The IUPAC name for uric acid is 7,9-Dihydro-1H-purine-2,6,8(3H)-trione. In its natural state, uric acid typically appears as a white crystalline substance or powder, possessing a molecular weight of 168 atomic mass units (amu).
Physical and Chemical Properties of Uric Acid
- Molecular weight: 168 amu.
- Appearance: White solid or powder.
- Odor: Odorless.
- Melting point: Greater than 300°C.
- Solubility: Highly soluble in water.
- pKa value: 5.4.
- Density: 1.87 g/cm³.
- Acidity: Classified as a weak organic acid.
- pH value: 7.4
Uses of Uric Acid
Disease Diagnosis: Monitoring the concentration of uric acid in the body is valuable for diagnosing various diseases, such as gout and kidney stones.
Inflammatory Response: Uric acid plays a role in initiating an inflammatory process, which is essential for tissue repair.
Immune System Stimulation: Urate, a form of uric acid, acts as a potent antioxidant and can stimulate the immune system.
Health Implications: High concentrations of uric acid in the body can lead to multiple health problems.
Control through Diet: Controlling uric acid levels can be achieved by increasing the intake of Vitamin C and maintaining proper hydration.
Elimination: Excess uric acid is expelled from the body through the process of urination.
Uric acid is a chemical compound composed of Carbon, Hydrogen, Nitrogen, and Oxygen, with a chemical formula of C 5 H 4 N 4 O 3 . It forms as a result of purine breakdown and is characterized by its white crystalline appearance and a molecular weight of 168 amu. Uric acid possesses various physical and chemical properties, including being odorless, highly soluble in water, and classified as a weak organic acid with a pH value of 7.4.
This compound serves essential roles in the body, including its use in diagnosing diseases like gout and kidney stones, its involvement in initiating inflammatory responses for tissue repair, and its function as an immune system stimulant due to its antioxidant properties. However, excessive uric acid levels can lead to health problems, which can be managed through dietary adjustments and adequate hydration. The body eliminates excess uric acid through the process of urination. Understanding uric acid and its properties is crucial for maintaining overall health and well-being.
Uric Acid Formula FAQs
What is uric acid?
What is the chemical formula of uric acid?
What is the IUPAC name of uric acid?
What is the appearance of uric acid?
How is uric acid eliminated from the body?










