
Physical Changes
Physical and chemical change of Class 7
Any change which can alters some physical property of the matter such as its state, texture, magnetic or electrical conditions or its colour, without causing any change in the composition of its molecules, is called physical change, or you can say it is a temporary change which can be easily reversed and in which only the physical properties of a substance change without any change in its composition. “Properties such as shape, size, colour and state of a substance are called physical properties. A change in which a substance undergoes a change in its physical properties is called a physical Change .
Following points need special consideration:
- Physical change is reversible and temporary.
- The composition of the substance does not change during a physical change and hence, no new substance is formed.
- No loss or gain of energy takes place during a physical change. The energy needed to bring about a physical change is equal to energy that can reverse the change. For example, 2260 J of heat energy is needed to change 1 g of water at 100°C to 1g of steam at 100°C. The same amount of heat energy is given out when 1g of steam condenses to 1 g of water at 100°C.
- No change in mass occurs during a physical change. This is because the substance undergoing a change remains the same. No matter is added or removed.
Examples of physical change:
- Making of ice-cream
- Boiling water into steam.
- Crystallization of sugar from its solution,
- Sublimation of camphor.
- Bending of glass tube by heating.
- Melting of wax.
- Evaporation of water.
- Glowing of an electric bulb
- Magnetising an iron bar by means of electricity.
- Dissolving sodium chloride in water

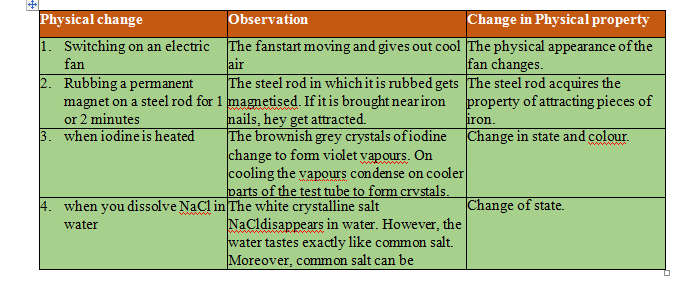
Some Examples Involving Physical Changes:


🔥 Trending Blogs
Talk to a counsellorHave doubts? Our support team will be happy to assist you!

Check out these Related Articles
Free Learning Resources
PW Books
Notes (Class 10-12)
PW Study Materials
Notes (Class 6-9)
Ncert Solutions
Govt Exams
Class 6th to 12th Online Courses
Govt Job Exams Courses
UPSC Coaching
Defence Exam Coaching
Gate Exam Coaching
Other Exams
Know about Physics Wallah
Physics Wallah is an Indian edtech platform that provides accessible & comprehensive learning experiences to students from Class 6th to postgraduate level. We also provide extensive NCERT solutions, sample paper, NEET, JEE Mains, BITSAT previous year papers & more such resources to students. Physics Wallah also caters to over 3.5 million registered students and over 78 lakh+ Youtube subscribers with 4.8 rating on its app.
We Stand Out because
We provide students with intensive courses with India’s qualified & experienced faculties & mentors. PW strives to make the learning experience comprehensive and accessible for students of all sections of society. We believe in empowering every single student who couldn't dream of a good career in engineering and medical field earlier.
Our Key Focus Areas
Physics Wallah's main focus is to make the learning experience as economical as possible for all students. With our affordable courses like Lakshya, Udaan and Arjuna and many others, we have been able to provide a platform for lakhs of aspirants. From providing Chemistry, Maths, Physics formula to giving e-books of eminent authors like RD Sharma, RS Aggarwal and Lakhmir Singh, PW focuses on every single student's need for preparation.
What Makes Us Different
Physics Wallah strives to develop a comprehensive pedagogical structure for students, where they get a state-of-the-art learning experience with study material and resources. Apart from catering students preparing for JEE Mains and NEET, PW also provides study material for each state board like Uttar Pradesh, Bihar, and others
Copyright © 2025 Physicswallah Limited All rights reserved.
Get App








