
Selenous Acid Formula : Selenium is an element denoted by the symbol Se and with an atomic number of 34. It belongs to the oxygen group and is classified as a non-metal. At room temperature, it exists in a solid state and exhibits properties that are intermediate between the elements located above and below it in the periodic table. Selenium plays a crucial role as a trace element and antioxidant and shares similar characteristics with sulfur, tellurium, and arsenic.
Oxygen, represented by the symbol O, possesses an atomic number of 8 and an electronic configuration of 1s²2s²2p⁴. It is a member of the chalcogen group within the periodic table and is known for its reactivity. Oxygen is commonly found in water, rocks, minerals, and a variety of organic compounds, making it the most abundant element in the Earth's crust.
Hydrogen, symbolized as H, is the lightest element with an atomic number of 1 and an electronic configuration of 1s¹. It holds the distinction of being the most abundant element in the universe. Each hydrogen atom contains just one proton and is typically found in a gaseous state.
Selenous Acid Formula
Selenous acid formula is H 2 SeO 3 , and it is a white, crystalline solid and serves as the primary oxoacid of selenium. It is predominantly used for the preservation and alteration of the color of steel, especially on firearm components. Additionally, it finds widespread application in the direct synthesis of various organoselenium derivatives and is the conjugate acid of a hydrogen selenite.
Selenous Acid Formula Structure
Selenous Acid, with the chemical formula H 2 SeO 3 , exhibits a structural arrangement more precisely described as (HO) 2 SeO, where selenium serves as the central atom. The structural representation of selenous acid is depicted below.
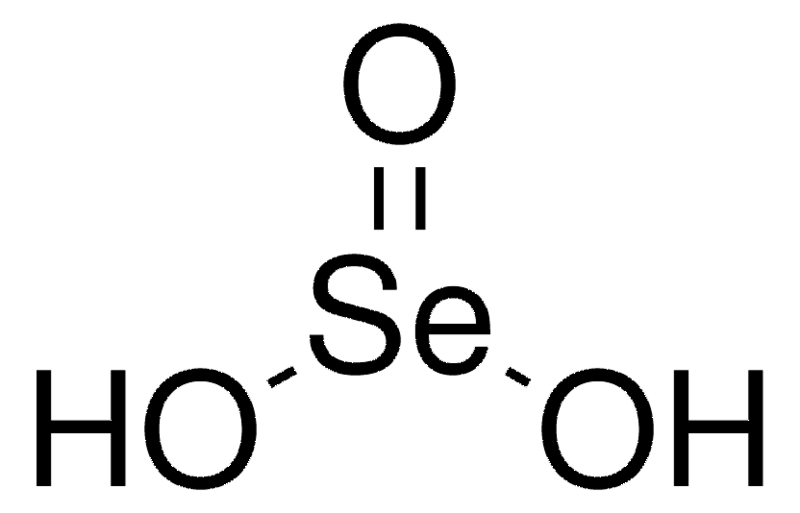
Preparation of Selenous Acid
Selenous Acid is synthesized through the following chemical reactions:
Selenium Dioxide reacts with water to yield selenous acid.
H 2 O + SeO 3 → H 2 SeO 3
Nitric acid interacts with selenium, resulting in the formation of selenous acid.
2 HNO 3 + Se → NO + NO 2 + H 2 SeO 3
The reaction between hydrochloric acid and Selenic acid produces chlorine, water, and Selenous Acid.
2 HCl + H 2 SeO 4 → H 2 O + Cl 2 + H 2 SeO 3
Barium selenate, when combined with hydrochloric acid, generates selenous acid.
4 HCl + BaSeO 4 → H 2 O + Cl 2 + BaCl 2 + H 2 SeO 3
Selenous Acid Formula Physical Properties
- Formula: Selenous Acid Formula is H 2 SeO 3
- Molecular Weight: 128.97 g/mol
- Density: 3.0 g/cm3
- IUPAC Names: Selenous Acid, Selenic(IV) Acid
- Solubility: Readily soluble in ethanol
- Melting Point: Decomposes at 70°C
Selenous Acid Formula Chemical Properties
Selenous Acid engages in various chemical reactions, as described below:
Reaction with Chloric Acid in Water yields chlorine and selenic acid.
2 HClO 3 + 5 H 2 SeO 3 → H 2 O + Cl 2 + 5 H 2 SeO 4
When Hydrogen Sulfide reacts with selenous acid, it results in the formation of water, sulfur, and selenium.
2 H 2 S + H 2 SeO 3 → 3 H 2 O + 2 S + Se
Interaction with hydrogen iodide produces water, iodine, and selenium.
4 HI + H 2 SeO 3 → 3 H 2 O + 2 I 2 + Se
In the presence of Sodium Hydroxide, selenous acid reacts to produce sodium selenite and water.
2 NaOH + H 2 SeO 3 → 2 H 2 O + Na 2 SeO 3
Uses of Selenous Acid
Selenous acid finds various practical uses, including:
Specialized Oxidizing Agent: Selenous acid serves as a specialized oxidizing agent in chemical processes.
Component for Gun Bluing: It is utilized as a component in gun bluing, a method for protecting and enhancing the appearance of firearm components.
Steel Color Modification: Selenous acid is used to protect and alter the color of steel, contributing to its aesthetic and protective properties.
Preservation of Vitamin E: Selenous acid plays a role in preserving vitamin E, an essential antioxidant in nutrition.
Synthesis of Organoselenium Derivatives: It is widely applied in the synthesis of diverse organoselenium compounds, extending its usefulness in chemical reactions.
Contribution to Dental Structure: Selenous acid is significant for its involvement in the structure of teeth, highlighting its role in dental health and development.
Nutritional Deficiency Treatment: In the realm of nutrition, selenous acid is used for the treatment of deficiencies, helping to restore vital selenium levels in the body.
| Related Links | |
| Gold Formula | Phosphate Formula |
| Helium Formula | Hydrazine Formula |
Selenous Acid Formula FAQs
What is the chemical formula of Selenous Acid?
What is the structure of Selenous Acid?
How is Selenous Acid prepared?
What are the physical properties of Selenous Acid?
What are the main chemical properties of Selenous Acid?










