A colorless, flammable liquid with disinfectant properties, propane-2-Ol is commonly used as an antiseptic. It is derived from 1-propanol and is used in pharmaceuticals, window cleaners, dye solutions, perfumes, cosmetics, antifreeze, lacquer formulations, and soaps. As rubbing alcohol, propane-2-Ol is sold at 70% aqueous concentration. As the temperature drops, propane-2-Ol becomes viscous and freezes at −89 °C.
Structure of Propan-2-ol
As an isopropyl group linked to a hydroxyl group, it is the simplest secondary alcohol.
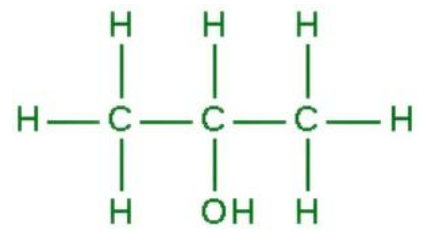
Propan 2 ol is a secondary alcohol formed by combining an isopropyl and hydroxyl groups. It shares similarities with other molecules, such as 1 propanol and ethyl methyl ether. With a molecular weight of 60.096 g mol -1 , it has a density of 0.786 g/cm3 at room temperature. Its boiling point is 82.6°C and its melting point is -89°C.
Properties of Propan-2-Ol
| Chemical formula | C 3 H 8 O |
| Molecular weight | 60.096 g·mol−1 |
| Density | 0.786 g/cm3 (20 °C) |
| Chemical names | s-Propanol, 2-Propanol, Isopropanol[1] Rubbing alcohol, iPrOH, sec-Propyl alcohol, i-PrOH, Dimethyl carbinol |
| Boiling point | 82.6 °C |
| Melting point | −89 °C |
Preparation of Propan-2-Ol
Biological process
Diabetic ketoacidosis produces very little propan-2-ol.
Hydration
By adding water or sulfuric acid to propene, these processes give primarily propane-2-ol, a secondary alcohol, rather than 1-propanol, a primary alcohol.
Also Check – Azelaic Acid Formula
Chemical Properties and Reactions
Oxidation
Chromic acid (H 2 CrO 4 ) oxidizes propan-2-ol to form acetone.
(CH 3 ) 2 CHOH ⇢ (CH 3 ) 2 CO + H 2 ⇡
Dehydration -
Using concentrated H 2 SO 4 at 443K or H 3 PO 4 at 440K, propane-2-ol is dehydrated, and propene and water are produced.
(CH 3 ) 2 CHOH ⇢ CH 2 =CH-CH 3 + H 2 O
Halogenation
Phosphorus tribromide is used to produce 2-bromopropane.
(CH 3 ) 2 CHOH ⇢ (CH 3 ) 2 CHBr
Also Check – Sugar Formula
Uses of Propanols
Because of its miscibility, strong flammability, and effects on the human body, propanol has a wide range of applications. These include:
As a Solvent
The majority of propanols are used as solvents and intermediates. In printing inks, particularly flexographic inks, propanol is used as a solvent. It is also added to floor polishes and metal degreasers in the cleaning industry. Adhesive makers use propanol as an extra solvent.
The coating industry uses propanol as a semi-volatile alcohol to improve drying properties, such as in alkyd resin paints, baking finishes, and electrodeposition paints.
There are also many other uses for propane including deicing fluids, extracting agents, and entrainers in azeotropic distillations. Propanol readily produces esters and ethers, the majority of which are highly valuable.
In addition to being used as a solvent, propanol is also used to produce other solvents such as antifreezes, lacquer formulations, soaps, dye solutions, window cleaning agents, and other compounds. In addition to making degreasing fluids, adhesives, and window cleaners, propanol is also used as an intermediate in the synthesis of halides, propyl amines, and propyl acetate.
Medicinal Uses
Also, propan-2-ol is used in a variety of pharmaceuticals. It is the main component in rubbing alcohol bottles. Additionally, it is an antiseptic. Propan-2-ol can also be found in hand sanitizer solutions or wipes. As an alternative to ethanol, propanol is frequently used in inhalant products, especially nebulizers.
It is used as a pain reliever since n-propyl alcohol can cause modest central nervous system depression.
The study published in Chem Pharm Bull in Tokyo in 1980 found that propanol triples the activity of human erythrocyte acid phosphatase (Eapase). In comparison to eapase, n-propanol activated human prostatic acid phosphatase (papase) less.
Propanol Tablet treats, controls, prevents, and improves supraventricular arrhythmias, ventricular tachycardias, digitalis-induced tachyarrhythmias, resistant tachyarrhythmias caused by excessive catecholamine activity during anaesthesia, and other conditions.
A number of foods and non-alcoholic beverages contain propanol in trace amounts as a volatile flavour.
Also Check – Hexane Formula
Cosmetics
Acetate is a common ingredient in cosmetics because of its moderate corrosive properties, which makes it an effective remover for acrylic nails and fingernail polish. It is also used in soaps, hair care products, perfumes, hand sanitizers, and moisturisers because of its moderate corrosive qualities.
Fuel
In addition to being highly flammable, propanol has a fire hazard rating of 2 and is also an excellent fuel for internal combustion engines due to its high octane number. However, the manufacture of propanol is too expensive to make it a popular fuel, therefore it is not often utilised for this purpose. When propane is used as a fuel, it has an anti-knock index (AKI) of 108 and a research octane number (RON) of 118.
Propanol is primarily used as a solvent, but other applications and uses of propanol are growing around the globe. Furthermore, propanol is less hazardous to use than other synthetic alcohols.
Propan-2-Ol Formula FAQs
Q1. What is the chemical formula of propan-2-ol?
Q2. What is the common name for propan-2-ol?
Q3. What are the physical properties of propan-2-ol?
Q4. What are the uses of propan-2-ol?
Q5. Is propane-2-ol safe to use?













