

Strontium Chloride Formula: Strontium chloride (SrCl 2 ) stands as a common salt, composed of both strontium and chlorine. This common halide, known as strontium chloride, plays a pivotal role in the creation of collodion chloride printing emulsions. it was the first toothpaste ingredient believed to block tubules, though this understanding stemmed from the widely misconstrued utilization of radiolabeled strontium and autoradiography. Notably, strontium compounds exhibit lower levels of toxicity.
Strontium Chloride Formula Structure
Strontium Chloride Formula is SrCl 2 . The structure of strontium chloride can be described as a crystalline solid, resembling the fluorite-like arrangement. In the gaseous phase, the SrCl 2 molecule takes on a non-linear form with a Cl-Sr-Cl angle of approximately 130 degrees. In this context, the VSEPR theory, which typically anticipates a linear structure, departs from the norm. Ab initio calculations have been invoked to argue that the d orbitals within the sub-valence shell hold sole responsibility for this deviation. Alternatively, some postulate that the electron core polarization of the strontium atom induces a distortion in the core electron density, directly influencing the Sr-Cl bonds.
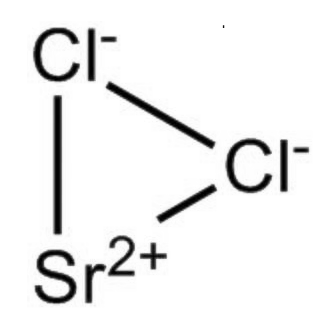
Strontium Chloride Formula Physical Properties
Chemical Formula: Strontium Chloride Formula is SrCl 2.
Odor: Odorless
Appearance: Appears as a white crystalline solid
Vapour Pressure: At 25°C, the vapour pressure is measured at 8.37 mm
Solubility: Strontium chloride is soluble in water and slightly soluble in alcohol.
Heat of Solution: The heat of solution is -7.50 calories.
Strontium Chloride Formula Chemical Properties
Strontium Chloride Formula is SrCl 2 . When strontium chloride (SrCl 2 ) reacts with fluorine gas (2HF), it forms chlorine gas (Cl 2 ) and strontium fluoride (SrF 2 ).
SrCl 2 + 2HF → SrF 2 + Cl 2
Strontium chloride (SrCl2) also reacts with sulfuric acid (2H 2 SO 4 ), resulting in the formation of hydrogen chloride (2HCl) and strontium hydrogen sulfate (Sr(HSO 4 ) 2 ).
SrCl 2 + 2H 2 SO 4 → Sr(HSO 4 ) 2 + 2HCl
Strontium Chloride Preparation
The process of preparing strontium chloride involves reacting either strontium carbonate or aqueous strontium hydroxide with hydrochloric acid, resulting in the following chemical equation:
Sr(OH)2 + 2 HCl → SrCl 2 + 2 H 2 O
The hexahydrate form SrCl 2 ·6H 2 O is obtained through crystallization from a cold aqueous solution. Dehydration occurs in several stages, starting at temperatures above 61°C (or 142°F), with complete dehydration occurring at 320°C (or 608°F).
Uses of Strontium Chloride
Strontium chloride finds various practical applications across distinct domains:
Strontium Chloride Formula is SrCl 2 . Strontium chloride plays an important role in the production of various strontium compounds like yellow strontium chromate, strontium carbonate, and strontium sulfate. When aqueous solutions of strontium chloride react with the sodium salt of the desired anion. It often leads to the formation of a solid precipitate.
SrCl 2 + Na 2 CrO 4 → SrCrO 4 + 2 NaCl
SrCl 2 + Na 2 CO 3 → SrCO 3 + 2 NaCl
SrCl 2 + Na 2 SO 4 → SrSO 4 + 2 NaCl
In the field of pyrotechnics, strontium chloride is commonly used as a red dye, creating notably brighter red flames compared to many other options. It's also utilized in small amounts in the manufacturing of glass and in metallurgy processes. Furthermore, for combating bone cancer, the radioactive isotope strontium-89 is administered in the form of strontium chloride. In the context of seawater aquaria, strontium chloride serves as an essential component, being consumed during the growth of specific plankton.
In the context of dental care, SrCl 2 is valuable for reducing tooth sensitivity by creating a protective barrier over tiny tubules in the exposed dentin, which contains nerve endings often revealed due to gum recession. While these products are known as "strontium chloride toothpastes" in the U.S., many now utilize saltpeter (KNO3) for its analgesic properties.
In biological research, brief exposure to strontium chloride can trigger parthenogenetic activation of oocytes, a phenomenon commonly utilized in developmental biological research.
For ammonium storage at low pressure, a commercial company employs a strontium chloride-based artificial solid named AdAmmine. This approach is primarily used for reducing NOx emissions in diesel vehicles. The company claims that their patented material can also be synthesized from other salts, but they have chosen strontium chloride for mass production.
Initially, the company considered using the stored ammonium for synthetic Ammonium fuel under the trademark HydrAmmine and the press name "hydrogen tablet," but this aspect has not been commercialized. Their processes and materials are protected by patents.
In soil testing, strontium chloride, in combination with citric acid, functions as a universal extractant for plant nutrients.
Side Effects of Strontium Chloride
Strontium, a non-radioactive silvery metal naturally found, predominantly accumulates in bones, constituting approximately 99% of its presence in the human body.
Distinct forms of strontium metal are being explored for medicinal purposes. Scientists are investigating the potential of strontium ranelate to be administered orally for the treatment of conditions like osteoporosis and arthritis, particularly concerning bone health.
| Related Links | |
| Barium Bromide Formula | Barium Fluoride Formula |
| Hydrobromic Acid Formula | Barium Iodide Formula |
Strontium Chloride Formula FAQs
What is the formula of strontium chloride?
What is the appearance of strontium chloride?
Does strontium chloride have a noticeable odor?
What level of vapor pressure does strontium chloride exhibit at 25°C?
Is strontium chloride soluble in water?












