
These charges build up from rubbing insulating surfaces together, a process known as static electricity. CBSE Class 12 Physics Notes for Chapter 1 Electric Charges and Fields explain these occurrences in detail. The chapter covers electrostatics, the branch of physics that studies forces, fields, and potentials resulting from static charges. It provides a thorough understanding of how and why these electrical phenomena happen, helping students grasp the underlying principles of electric charges and fields.
CBSE Class 12 Physics Notes Chapter 1 Electric Charges and Fields Overview
These notes are prepared by experts of Physics Wallah give a clear overview of CBSE Class 12 Physics Chapter 1 Electric Charges and Fields. They explain important ideas like static electricity and how electric charges can create sparks or lightning. The notes cover how these charges build up and discharge, leading to various phenomena.CBSE Class 12 Physics Notes Chapter 1 Electric Charges and Fields PDF
The PDF for CBSE Class 12 Physics Notes Chapter 1 Electric Charges and Fields is available below. It provide valuable insights into the fundamental principles of electrostatics helping students understand and review the chapter efficiently. Click the link below to access and download the PDF for your study needs.CBSE Class 12 Physics Notes Chapter 1 Electric Charges and Fields PDF
CBSE Class 12 Physics Notes Chapter 1 Electric Charges and Fields
Here are the notes for CBSE Class 12 Physics Chapter 1 Electric Charges and Fields. This chapter explains the basics of electric charges and the electric fields they create. It includes key concepts like Coulomb's Law, which describes the force between two charged objects, and the superposition principle, which helps calculate the total electric field from multiple charges. You'll also learn about electric field lines, their properties and electric flux. These concepts are essential for understanding how electric forces work. You can find the full notes through the link provided for more details.Electric Charge
The term 'electricity' comes from the Greek word "Elektron," which means amber. This is because the ancient Greeks discovered that rubbing amber would generate static electricity. In physics, electric charge is a fundamental property of matter, influencing how atoms and molecules interact. There are two types of electric charge: positive and negative. The fundamental rule of electric charges is that like charges repel each other, while unlike charges attract. This property is known as the polarity of charge and is key to understanding many electrical phenomena.Conductors and Insulators
In experiments with electric charges, it was discovered that conductors and insulators behave very differently. Conductors, such as metals, the Earth, and the human body, allow electric charges to move freely through them. This means that they can easily carry electricity. On the other hand, insulators, like porcelain, nylon, and wood, resist the flow of electric charges. They do not conduct electricity well, making them useful for preventing unwanted electrical flow and protecting us from electric shocks. Read More - Strategy to Solve Class 12 Sample Papers EfficientlyProperties of Electric Charge
Electric charge has three key properties: Quantization : This property indicates that the total charge on a body is an integer multiple of a basic unit of charge. This means that electric charge comes in discrete amounts rather than continuous values. Additivity : The total charge in a system is the algebraic sum of all individual charges present. This means that if multiple charges act on a system, their effects add together to give the overall charge. Conservation : This principle states that the total charge in an isolated system remains constant over time. Charges cannot be created or destroyed, only transferred from one object to another. This ensures that the total charge of a system is always conserved.Coulomb's Law
Coulomb's law describes the electrostatic force between two point charges. If two stationary point charges Q1 and Q2 are placed a distance r apart, the magnitude of the electrostatic force F between them is given by: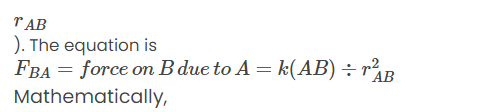 Key Points: Direction of Force : The force acts along the line joining the two charges. Nature of Force :
Key Points: Direction of Force : The force acts along the line joining the two charges. Nature of Force :
- Repulsive : If the charges are of the same sign (both positive or both negative).
- Attractive : If the charges are of opposite signs (one positive and one negative).
Forces Between Multiple Charges
When dealing with multiple charges, Coulomb's law applies to the interaction between any two charges. However, calculating the net force on a specific charge in the presence of several other charges requires a different approach. The net force on a charge due to multiple other charges is determined by taking the vector sum of all the individual forces exerted on that charge by each of the other charges, considered one at a time. This principle is known as the superposition principle .Superposition Principle
The superposition principle states that the interaction between any two charges is not influenced by the presence of other charges. In simpler terms if you have multiple charges in a system the force between any two specific charges remains the same, regardless of other charges in the vicinity.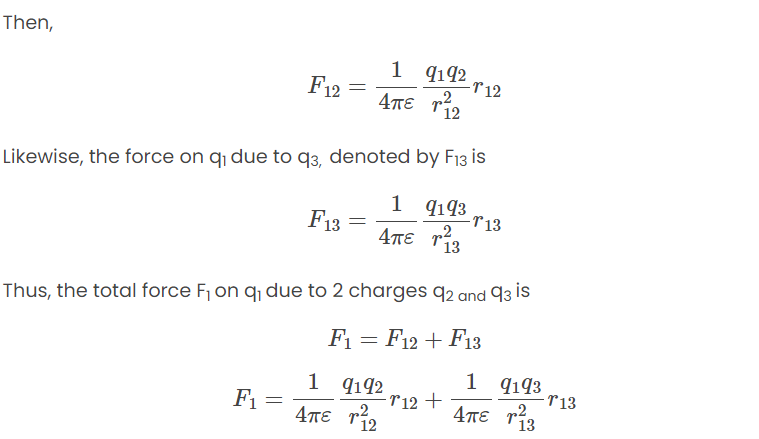
Properties of Electric Field Lines
Electric field lines have several key properties that help visualize the behavior of electric fields: Continuous Curves : Electric field lines form continuous curves without any breaks in regions where there are no charges. This continuity shows the direction of the field at every point in space. No Intersection : Two electric field lines never cross each other. If they did, it would imply that the electric field has two different directions at the same point, which is impossible. Starting and Ending Points : Electric field lines originate from positive charges and terminate on negative charges. This indicates the direction of the force that a positive test charge would experience in the field. No Closed Loops : Electrostatic field lines never form closed loops. They always start on a positive charge and end on a negative charge, or they extend to infinity if no negative charge is present. This distinguishes electric fields from magnetic fields, where lines can form closed loops.Electric Flux
Electric flux refers to the total number of electric field lines passing through a given area. It doesn't involve a physical flow like liquids but is a measure of how strong the electric field is over that area. The electric flux Δθ through a small area element ΔS is given by:Δθ= E.ΔS= E ΔS cosθ
Here:- E is the electric field strength,
- ΔS is the area element,
- θ is the angle between the electric field E and the normal (perpendicular) to the area element ΔS
Electric Dipole
An electric dipole is a system of two equal and opposite charges, typically denoted as +q and −q separated by a certain distance. The dipole moment is calculated as the product of the magnitude of one of the charges and the distance separating them, mathematically expressed as:p = q × 2 a
Here, 2a represents the distance between the charges, and the direction of the dipole moment vector is from the negative charge to the positive charge. The concept of an electric dipole is important in understanding how molecules and other systems interact with electric fields.Charge on a Capacitor
The net charge on a capacitor is always zero because the charges on its two plates are equal in magnitude but opposite in sign. However, when we talk about the charge Q on a capacitor, we refer to the magnitude of the charge on one of the plates.Properties of an Ideal Battery
An ideal battery is a theoretical model that simplifies the behavior of real batteries by assuming certain ideal characteristics. The properties of an ideal battery include: Two Terminals :- A battery has two terminals: one positive and one negative.
- The potential difference V between the terminals of the battery remains constant regardless of the load connected to it.
- The terminal with the higher potential is the positive terminal, and the terminal with the lower potential is the negative terminal.
- The fixed potential difference between the terminals of the battery is referred to as the electromotive force (emf) of the battery.
- When a conductor is connected to a terminal of the battery, the potential of the conductor matches the potential of the terminal.
- If the plates of a capacitor are connected to the terminals of the battery, the potential difference across the capacitor becomes equal to the emf of the battery.
- The total charge within the battery remains zero. If the positive terminal of the battery supplies a charge Q the negative terminal will supply an equal and opposite charge −Q.
- When a charge Q moves from the negative terminal to the positive terminal through the battery, the battery does work QE , where E is the emf of the battery.
- An ideal battery is often represented by a symbol with two parallel lines of unequal length. The longer line represents the positive terminal, and the shorter line represents the negative terminal.
- The potential difference between these lines is equal to the emf of the battery.
Benefits of CBSE Class 12 Physics Notes Chapter 1 Electric Charges and Fields
Clear Conceptual Understanding : The notes provide a simplified explanation of fundamental concepts such as electric charges, Coulomb's law, and electric fields, making complex topics easier to understand. Concise Summary : They concise information from the textbook into a manageable format, allowing students to review key points quickly and efficiently. Enhanced Retention : By highlighting essential concepts and formulas, these notes aid in better retention of important details needed for exams. Effective Revision Tool : During exam preparation these notes are a quick reference to revise the topics, saving time compared to reading through entire chapters. Comprehensive Coverage : The notes cover all key aspects of the chapter including definitions, laws, properties, and applications, providing a thorough review of the topic.CBSE Class 12 Physics Notes Chapter 1 Electric Charges and Fields FAQs
What is an electric charge?
An electric charge is a fundamental property of matter that causes it to experience a force when placed in an electromagnetic field. There are two types of electric charges: positive and negative.
What are the types of electric charges?
There are two types of electric charges: positive (e.g., protons) and negative (e.g., electrons). Like charges repel each other, while unlike charges attract each other.
What is the principle of superposition in electric fields?
The principle of superposition states that the net electric field at a point due to multiple charges is the vector sum of the electric fields due to each charge taken individually.
What is an electric field?
An electric field is a region around a charged object where other charges experience a force. It is defined as the force per unit charge and is represented by field lines that point away from positive charges and toward negative charges.
What are the properties of electric field lines?
Electric field lines are continuous curves without breaks, never intersect, start at positive charges and end at negative charges, and do not form closed loops.
🔥 Trending Blogs
Talk to a counsellorHave doubts? Our support team will be happy to assist you!

Check out these Related Articles
Free Learning Resources
PW Books
Notes (Class 10-12)
PW Study Materials
Notes (Class 6-9)
Ncert Solutions
Govt Exams
Class 6th to 12th Online Courses
Govt Job Exams Courses
UPSC Coaching
Defence Exam Coaching
Gate Exam Coaching
Other Exams
Know about Physics Wallah
Physics Wallah is an Indian edtech platform that provides accessible & comprehensive learning experiences to students from Class 6th to postgraduate level. We also provide extensive NCERT solutions, sample paper, NEET, JEE Mains, BITSAT previous year papers & more such resources to students. Physics Wallah also caters to over 3.5 million registered students and over 78 lakh+ Youtube subscribers with 4.8 rating on its app.
We Stand Out because
We provide students with intensive courses with India’s qualified & experienced faculties & mentors. PW strives to make the learning experience comprehensive and accessible for students of all sections of society. We believe in empowering every single student who couldn't dream of a good career in engineering and medical field earlier.
Our Key Focus Areas
Physics Wallah's main focus is to make the learning experience as economical as possible for all students. With our affordable courses like Lakshya, Udaan and Arjuna and many others, we have been able to provide a platform for lakhs of aspirants. From providing Chemistry, Maths, Physics formula to giving e-books of eminent authors like RD Sharma, RS Aggarwal and Lakhmir Singh, PW focuses on every single student's need for preparation.
What Makes Us Different
Physics Wallah strives to develop a comprehensive pedagogical structure for students, where they get a state-of-the-art learning experience with study material and resources. Apart from catering students preparing for JEE Mains and NEET, PW also provides study material for each state board like Uttar Pradesh, Bihar, and others
Copyright © 2025 Physicswallah Limited All rights reserved.
Get App









