science class 10th chapter-Carbon and its compound Formula & Important points
May 13, 2022, 16:45 IST
Class 10th for chapter-Carbon and its compound
This sheet of pdf consist of short notes, formula , important bullet points of science class 10th prepared by expert faculty members of Physics Wallah for quick recap of the entire chapter - Carbon and its compound of class 10 science. This pdf sheet of chapter Carbon and its compound is useful for last minutes revision before exam . If you required detail science class 10th then visit class 10 science section. To score good marks in class 10 solve questions from NCERT with the help of NCERT solutions . Physics Wallah expert uploaded NCERT solutions for class 10 Science .
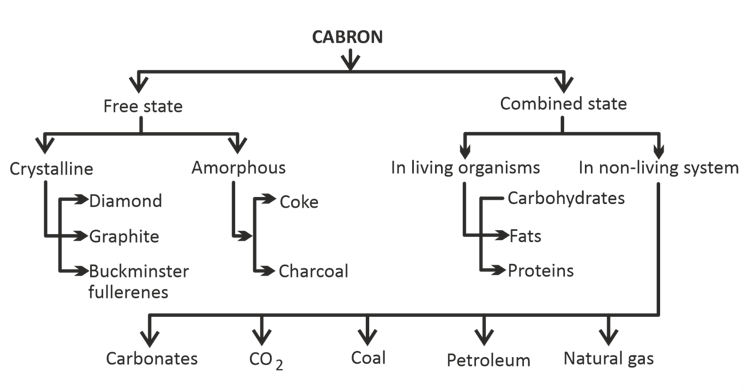
Difference between Diamond and Graphite
| S.NO. | Diamond | S.NO. | Graphite |
| 1. | Diamond has a three-dimensional network structure. | 1. |
Graphite has a two-dimensional sheet like
structure consisting of a number of benzene rings fused together. |
| 2. | It is the hardest natural substance known. | 2. |
Graphite is soft and greasy and is used as
solid lubricant for heavy machinery operating at high temperatures. |
| 3. |
It is a bad conductor of electricity but is
a very good conductor of heat. Because of hardness and high thermal conductivity, diamond tipped tools do not overheat and hence are extensively used for cutting and drilling purposes. |
3. |
It is a good conductor of both heat and electricity.
Because of high electrical conductivity, graphite is used for making electrodes of battery and arcs. |
| 4. |
It s a transparent substance with high refractive index.
Therefore, it is used for making gemstones and jewellery. |
4. | It is an opaque grayish black substance. |
Simplest compounds of carbon are Hydrocarbons(compounds having atoms of carbon and hydrogen only)
For additional information related to the subject you can check the Chemistry Formula section.
| Name of Alkanes | Molecular formula | Condensed Structural Formula |
| Methane | CH 4 | CH 4 |
| Ethane | C 2 H 6 | CH 3 CH 3 |
| Propane | C 3 H 8 | CH 3 CH 2 CH3 |
| Butane | C 4 H 10 | CH 3 CH 2 CH 2 CH3 |
| Pentane | C 5 H 12 | CH 3 CH 2 CH 2 CH 2 CH 3 |
| Hexane | C 6 H 14 | CH 3 CH 2 CH 2 CH 2 CH 2 CH 3 |
| Heptane | C 7 H 16 | CH 3 CH 2 CH 2 CH 2 CH 2 CH 2 CH 3 |
| Octane | C 8 H 18 | CH 3 CH 2 CH 2 CH 2 CH 2 CH 2 CH 2 CH 3 |
| Nonane | C 9 H 20 | CH 3 CH 2 CH 2 CH 2 CH 2 CH 2 CH 2 CH 2 CH 3 |
| Decane | C 10 H 22 | CH 3 CH 2 CH 2 CH 2 CH 2 CH 2 CH 2 CH 2 CH 2 CH 3 |
Download Free Pdf sheet of Carbon and its compound it consist of Short notes and key points of Chemistry for class 10 from the link given below.











