SSC Worksheet for chapter-1 Chemical Reactions class 10
Worksheet For class 10
This page is prepared by the Academic team of Physics Wallah which consists of SSC Board Worksheet for Class 10 Chemistry. Students of Class 10 Chemistry can get a free Worksheet for Class 10 Chemistry in PDF format prepared as per the newest syllabus and examination pattern in your schools.
Before solving the Worksheet for class 10 one must read the textbook of your school board exam and try to read the notes given in Physics Wallah class 10 notes sections. Students can also download free pdf of Class 10 Chemistry Notes prepared by teachers and solve important problems provided here with solutions on daily basis to get more scores in school exams and tests.
Physics Wallah worksheets for class 10 SSC can help students practice with tests and quickly revise lessons learned in the classroom with essential resources such as video lessons, short notes, and sample papers.
Class 10 Board is one of the most important year for every student and need a good planning and right study material to score good marks in your final exam. If any students need to take the online test to check their concepts or undertstanding then they can visit Chemistry Quiz for Class 10.
Objective Type Questions
Q1. Fe2O3 + 2Al →Al2O3 + 2Fe
The above reaction is an example of a
- Combination reaction
- Double displacement reaction
- Decomposition reaction
- displacement reaction
Q2. Which of the following is not a physical change ?
- Boiling of water to give water vapour
- Melting of ice to give water
- Dissolution of salt in water
- Combustion of liquefied petroleum gas
Q3. 4NH3(g) + 5O2 (g)→4NO(g) + 6H2O(g)
Which type of reaction is this ?
- Displacement reaction
- Combination reaction
- Redox reaction
- Neutralisation reaction
Q4. Which of the following is/ are exothermic processes ?
- Reaction of water with quick time
- Dilution of an acid
- Evaporation of water
- Sublimation of camphor
Q5. Mole ratio of hydrogen and oxygen gases liberated during electrolysis of water is
- 1 : 1
- 2 : 1
- 4 : 1
- 1 : 2
Q6. Which of the following is/are an endothermic processes ?
- Dilution of sulphuric acid
- Sublimation of dry ice
- Condensation of water vapours
- Evaporation of water
Q7. Which one of the following processes involve chemical reactions ?
- Heating copper wire in the presence of air at high temperature
- Liquefaction of air
- Storing of oxygen gas under pressure in a cylinder
- Keeping petrol in open in a china dish
Q8. Which of the following are combination reactions ?
- Zn + FeSO4→ZnSO4 + Fe
- MgO + H2O→Mg(OH)2
-
2KClO3
.jpg) 2KCl + 3O2
2KCl + 3O2 - 4Al + 3O2→2Al2O3
Q9. Which of the following gas can be used for storage of fresh sample of an oil for a long time ?
- CO2
- N2
- He
- O2
Q10. Which among the following is /are double displacement reaction ?
- Pb + CuCl2→PbCl2 + Cu
- Na2SO4 + BaCl2→BaSO4 + 2NaCL
- C + O2→CO2
- CH4 + 2O2→CO2 + 2H2O
Q11. Write correct explanation for following reaction ?
2KMnO4 + FeSO4 + 8H2SO4→5Fe2 (SO4)3 + K2SO4 + 2MnSO4 + 8H2O
- FeSO4 acts as an oxidizing agent and oxidizes KMnO4
- KMnO4 acts as an oxidizing agent and oxidizes FeSO4
- H2SO4 acts as an oxidizing agent and oxidizes FeSO4
- H2SO4 acts as an oxidizing agent and oxidizes KMnO4
Q12. CaO(s) + H2O(l)→Ca(OH)2 + q
Which of the following statements is/are correct ?
- It is an endothermic reaction
- It is an exothermic reaction
- The pH of the resulting solution will be more than seven
- The pH of the resulting solution will be less than seven
Q13. BaCl2 + (NH4)2SO4→BaSO4↓+ 2NH4Cl
Which of the following is true for this reaction ?
- Displacement reaction
- Precipitation reaction
- Combination reaction
- Double displacement reaction
Q14. 2KClO3(s) 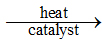 2KCl(s) + 3O2(g)
2KCl(s) + 3O2(g)
Which of the following statement(s) is /are correct about the reaction ?
- It is decomposition reaction and endothermic in nature
- It is combination reaction
- It is decomposition reaction and exothermic in nature
- It is photochemical decomposition and exothermic
Q15. Correct states of the reactions and products involved at reaction temperature ?
- 2H2 (l) + O2 (l)→2H2O (g)
- 2H2 (g) + O2 (l)→2H2O (l)
- 2H2 (g) + O2 (g)→2H2O (l)
- 2H2 (g) + O2 (g)→2H2O (g)
Subjective Type Questions
Q16. Identify the reducing agent in the following reactions.
- 4NH3 + 5O2→4NO +6H2O
- H2O + F2→HF + HOF
- Fe2O3 + 3CO→2Fe + 3CO2
- 2H2 + O2→2H2O
Q17. Identify the oxidizing agent on the following reactions.
- Pb3O4 + 8HCl→3PbCl2 + Cl2 + 4H2O
- 2Mg + O2→2MgO
- CuSO4 + Zn→Cu + ZnSO4
- V2O5 + 5Ca→2V + 5CaO
- 3Fe + 4H2O→Fe3O4 + 4H2
- CuO + H2→Cu + H2O
Q18. Grapes hanging on the plant do not ferment but after being plucked from the plant can be fermented under what conditions do these grapes ferment ? It is chemical or a physical change ?
Q19. Explain the following terms.
- Rancidity
- Endothermic reaction
- Redox reactions
- Neutralisation
- Corrosion
Q20. Oil and fat containing food items are flushed with nitrogen, why ?
Answer Key : Chemical Reactions
|
1. (d) |
2. (d) |
3. (c, d) |
4. (a, b) |
5. (b) |
6. (b, d) |
7. (a) |
8. (b, d) |
9. (c) |
10. (b) |
|
11. (b) |
12. (b, c) |
13.(b, d) |
14. (a) |
15. (c) |
|
||||
Solutions :-
Ans 16. In any reaction, reducing agent is the substance that undergoes oxidation.
- NH3 is the reducing agent because it is undergoing oxidation in the reaction
- F2
- CO
- H2, reason is the same for all
Ans 17. Oxidising agent is the substance that itself gets reduced in the reaction. It is the substance from which oxygen is removed or hydrogen is added.
- Pb3O4
- O2
- CuSO4
- V2O5
- H2O
- CuO
Ans 18. The fermentation of grapes takes place in the presence of anaerobic conditions ( in the absence of air). Since, new products are formed and it is an example of reversible reaction, this is a chemical change.
Ans19. (a) Rancidity→it is the chemical decomposition of fats, oils and other lipids. These chemical reactions results in undesirable odors and flavours. In this process their smell and taste gets change, usually substances which prevent oxidation (antioxidants) are added to foods containing fats and oils.
(b) Endothermic Reactions→Chemical reaction in which heat is absorbed by reactions
In endothermic reaction energy of products is greater than the energy of reactants.
Example - 2HgO + Heat→2Hg(l) + O2
N2(g) + O2(g) + Heat→2NO(g)
(c) Redox Reactions→It is a chemical reaction in which both oxidation and reduction both takes place. Oxidation is loss of electrons while reduction is gain of electrons.
Example - H2 + F2→2HK
Oxidation of hydrogen and reduction of fluorine is occurring
(d) Neutralisation ® The reaction between an acid and a base is called neutralization reaction. In this reaction, water and salt is formed.
Acid + Base→Salt + Water
H-A + BOH→BA + H2O
e.g. NaOH + HCl→NaCl + H2O
(e) Corrosion ® The process of slow degradation or eating up of metals when exposed to moist air is called combustion.
e.g. The black coating on silver and the green coating on copper are other examples of corrosion.
Silver develops black coat due to hydrogen sulphide of air
2Ag + H2S→Ag2S + H2
Ans 20. Oil and fat containing items when exposed to oxygen of air, get oxidized to form compounds that change the taste and smell of these food stuffs making them rancid.
Flushing this food item with nitrogen which is an inert gas protects the food items from getting rancid. The ‘Lays’ chips that you eat has nitrogen gas flushed in it to prevent rancidity.
Related Link
- Class 10 Physics Notes
- Class 10 Chemistry Notes
- Class 10 Biology Notes
- Class 10 Maths Notes
- Chapter Wise Quiz Class 10 Physics
- Chapter Wise Quiz Class 10 Chemistry
- Chapter Wise Quiz Class 10 Biology
- Chapter Wise Quiz Class 10 Maths
- Previous year Board Papers
- Sample Papers Maths Class 10
- Sample Papers Science Class 10
- NCERT Maths Solutions Class-10









