Alternative Or Non-Conventional Sources Of Energy
Source Of Energy of Class 10
Alternative Or Non-Conventional Sources Of Energy
Our energy demand is increasing day by day as our lifestyles are also changing. As our demand for energy increases, we need to look for more and more sources of energy. So technologies are developed to use the available or known sources of energy more efficiently and also look for new sources of energy.
SOLAR ENERGY:
The sun is the primary source of energy for all living beings on the earth. It provides all of us with heat and light. The energy generated by the sun is the result of a reaction called nuclear fusion, occurring continuously in the interior part of the sun. Hans Bethe, a physicist proposed that the enormous release of energy from the sun is due to the fusion (combination) of four hydrogen atoms to yield a single helium atom ( 24He).
For this discovery, he was awarded the 1967 Noble Prize in Physics. The sun emits energy in all directions in space.
Solar energy is trapped by plains, plateaus, mountains, rivers, lakes, oceans, and ponds. Plants use solar energy to manufacture food by photosynthesis and also solar energy is the source of wind storms, rain, snowfall & ocean waves.
It is the perennial (forever) source of energy.
This perennial source of energy has some features:
(i) It comes to earth surface in a very diffused form.
(ii) Upper atmosphere gets 1.3 KJ of energy per second per square meter of this only 47% i.e. approximately 0.64 KJ energy reaches per second per square meter on earth's surface.
(iii) Moreover this amount of energy is not available uniformly and keeps changing every day a place to place during the day.
(a) Composition of Solar Energy (Sunlight) :
Ultra violet rays:
The invisible rays whose wavelength is shorter than that of the visible violet light are called ultraviolet rays. We can detect these rays by using a photographic film or a fluorescent paper because they darken the photographic film just like ordinary light rays. They are used to kill bacteria in food and drinking water. Too much ultraviolet radiation is dangerous for our health due to its ionizing effect and can cause skin cancer.
Visible light :
The visible rays whose wavelength range from 400 nm (in violet) to 700 nm (in red) are called visible light The visible part of the sunlight consists of seven different wavelengths, each wavelength corresponding to a different color. Thus the visible part of the sunlight consists of seven different colors.
Infra-red rays :
The invisible rays whose wavelength is longer than that of the visible red light are called infra-red rays. They can heat the object on which they fall. About one-third of the solar energy consists of infra-red rays. They can be detected by their heating effect, by using a thermometer. Every hot object emits infra-red rays. They are used to get relief from body aches.
Uses of Solar Energy :
- Solar cooker absorb solar energy and cook food.
- Solar water heaters are used for heating water.
- Solar cells convert solar energy into electricity to run watches, calculators, and spaceships for various experiments.
- Solar energy is absorbed by green plants to make their food by photosynthesis.
- Solar energy is used for drying clothes and food grains.
- Solar energy is used for making salt from sea water.
Advantages of Solar Energy :
- It is inexhaustible and renewable as it is being produced continuously in the core of the sun by nuclear fusion of H-atoms.
- Its quantity is unlimited and is available in all parts of the world in abundance.
- It does not cause any pollution.
- It can be put to practical applications.
Limitations of Solar Energy :
- It is not available at night.
- It is not available uniformly in all parts of world.
- Solar energy received by the earth is quite diffused and in scattered form and hence only a part of it is utilized.
- It is not available at constant rate due to clouds, fog, mist, haze, winds etc.
Direct and Indirect Harnessing (or Collection) of Solar Energy:
- Direct utilization of solar energy can be done by collecting the heat radiated by reflecting these by plane mirrors onto black boxes containing uncooked food (in solar cooker) and for heating water in solar heaters. These rays can be converted into electrical energy as in solar cells.
- Indirect utilization of solar energy can be done by first converting solar energy into chemical energy as in biomass of plants. The heat energy of the sun can be utilized in sea waves (ocean thermal energy) and in the energy of winds etc.
Solar Heating Devices:
These are the devices that can collect and store heat obtained from solar energy. These are used for heating and cooking purposes. Solar heating devices are designed in such a way that they can make maximum utilization of solar heat radiations. It is done by adopting the following procedure:
- Concentration of solar energy by using reflectors: For moderate heating sun rays are reflected by using plane mirrors, as in solar cookers and solar water heaters. For high temperatures, sun's energy is concentrated using concave mirrors as reflectors.
- Black paint: Since black bodies are good absorbers as well as good radiators of heat, hence black paint is used to absorb and store heat radiations in large quantities by using a large surface area.
- Glass-sheet cover: Glass sheet cover is used to protect the hot infrared rays of solar energy from escaping the body of the black box. It allows the IR radiations (of shorter wavelength) to enter the box of the solar heating device and do not allow IR radiations (of longer wavelength) to escape from the solar heating device. Hence, more heat is retained by solar heating devices for a long time.
Box type solar cooker:
A solar cooker is a device that is used to cook food by utilizing the heat radiation coming from the sun. There are two types of solar cookers:
A box type solar cooker is based on the following facts:
Principle
A box – type solar cooker is based on the principle of the greenhouse effect. Glass reflects infrared radiations of longer wavelengths but allows those with shorter wavelengths to pass through. The solar radiation which passes through the glass heats the surfaces inside the box. These warm surfaces then radiate heat. This radiation consists mainly of IR of longer wavelengths. They are reflected by the glass. This process of heating is called the greenhouse effect.
Construction
- Outer wooden box (O) with thermo Cole lining – It prevents the loss of heat generated by greenhouse effect from the box.
- Inner black painted metal box (I) – A black body is a good absorber of the heat radiation. The blackened surface absorbs the maximum amount of heat (98%) and prevents heat loss due to reflection.
- Reflector (Plane Mirror), R – It increases the effective area for the collection of solar energy.
- Glass cover plate (P) – The purpose of glass cover plate is to trap heat radiations. It transmits 50% of heat radiation when it is coming from a source like the sun which is at a high temperature. But, when the radiations come from a source which is at a lower temperature, glass is opaque to them i.e., it does not allow the radiations to pass through it. Thus, the glass cover placed on the box produces a kind of green house effect.
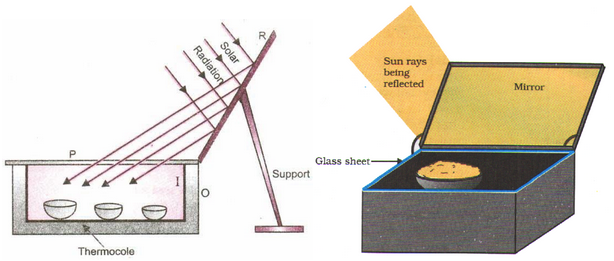
Working:
The sunlight falls on the plane mirror (reflector) which concentrates the light on the glass cover plate. The cover plate (P) heats the metal box through the greenhouse effect. This heat is conserved by the black paint and outer wooden box (O).
Uses:
The temperature inside the cooker can go upto 100°C – 140°C when kept in the sun for two to three hours. These cookers are provided with small containers so that 2 – 3 items can be cooked simultaneously.
(b) Merits and demerits of solar cookers
The various merits and demerits of solar cookers are as follows:
Merits
- Solar cookers do not require fuel and hence cause no pollution.
- The nutritional value of the food remains intact.
- Maintenance cost is low.
Demerits
- Food cannot be cooked at night or during cloudy days.
- Cooking takes more time.
Spherical reflector type solar cooker:
In this type of solar cooker, there is an insulated metal box, painted black from the inside. A spherical reflector is used here (in place of the plane mirror) because a very high temperature is required, the spherical reflector is either a concave reflector or a parabolic reflector. The sunlight falling on the surface of the spherical reflector gets concentrated at one point. This produces a lot of heat at that point & the temperature of that region becomes very high, therefore a utensil is placed at that point. Since a high temperature can be produced, so it can be used for making chappatis and for frying purposes.

Difference between box - type & spherical reflector type solar
|
Box -type solar cooker |
Spherical reflector type solar cooker |
||
|
1. |
A plane mirror reflector is used. |
1. |
Spherical reflector is used |
|
2. |
In a box type solar cooker, comparatively low temperature is produced |
2. |
Quite high temperature is produced in the spherical reflector type solar cooker. |
|
3. |
It cannot be used for making chappaties |
3. |
It can be used for frying and making chappaties. |
|
4. |
Used for cooking food requiring slow heating |
4. |
Used for cooking food requiring strong heating |
Solar Cell: A solar cell is a device which converts solar energy directly into electricity.
Thus, the limitations of using solar energy is overcome by using solar cells. It was in the year 1954 when the first practical solar cell, which could convert about 1% of the solar energy into electric energy, was fabricated. A typical solar cell consists of a 2 cm square piece of almost pure silicon can develop a voltage of 0.5v – 1v and can produce about 0.7 W of electricity when exposed to the sun.
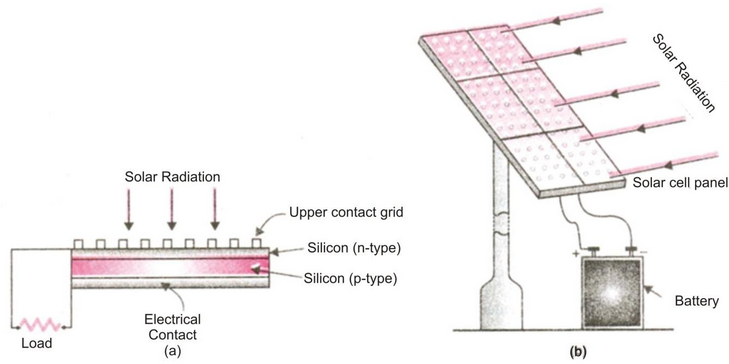
A solar cell
Semi-conductors:
Semi-conductors are those substances which have very low electrical conduciivity. Under ordinary conditions, semi-conductor materials conduct only a small amount of electric current. But if certain impurities are added to semi-conductor materials then their electrical conductivity increases considerably. Semi-conductors are neither good conductor of electricity nor they are completely insulators. The process of adding impurity is called doping. The material (semiconductors) doped with boron has an affinity to attract electrons and is termed as p-type (acceptor) semiconductor. The phosphorous doped silicon material, which has a surplus of electrons, is termed as n-type (donor) semiconductor. When solar energy fails on semi-conductor material, even then their electrical conductivity increases.
Conduction of solar cell :
It is constructed usually from silicon & gallium. Its conductivity increases when light falls on it. Therefore in a solar cell, the pieces (usually wafers) of semi conducting materials containing impurities are so arranged that when light falls on them then a potential difference is produced between two regions of the semi conductor. It has been observed that a solar cell of about 4 cm2 may produce potential difference of about 0.4V to 0.5V and generate a current of 60 mA. A large number of solar cells joined together in a definite pattern can provide much higher power for many use. The group of solar cells is called solar cell panel.
Solar cell panel:
When solar cells are arranged side by side, connecting each other in such a way that total potential difference and the total capacity to provide electric current is increased to a large extent the arrangement is called solar cell panel. The electric power required for the working of artificial satellites in outer space, street lighting in remote areas and running of irrigation water pumps in far-off areas obtained with the help of solar cell panels. In a solar cell panel hundreds of solar cells are joined together, the electricity produced by this solar panel is stored in battery. This battery runs an electric motor and finally the motor drives the water pump, which pumps out the underground water. The various solar cells in a solar cell panel are joined together by using connecting wires made of silver. This is because silver metal is the best conductor of electricity having very low resistance. The use of silver for connecting solar cells makes the solar cell panel more expensive but it increases the efficiency. This is because if connecting wires of other metals were used in solar cell panel, then a substantial part of the electricity generated by it could be lost in overcoming the resistance of such connecting wires.

Advantages of solar cells/ solar cell panels
- They have no moving parts, require little maintenance and work quite satisfactorily without the use of any focusing device.
- They can be set up in remote and inaccessible areas in which lying of power transmission line may be expensive and not commercially viable.
- Silicon which is used in solar cells is available abundantly and is also ecofriendly.
Disadvantages
- Special grade silicon for making solar cells is limited.
- The entire process of manufacture is still very expensive. Silver used for interconnection of the cells in the panel further adds to the cost.
Uses of solar cell:
- Solar cells are used for providing electricity in artificial satellite and space probes.
- In India, solar cells are being used for street lighting, for traffic signals, for operating water pumps and for running radio and television sets in remote areas.
- Solar cells are used for providing electricity to "lighthouses" situated in the sea and to the off-shore oil drilling rig platforms.
- Solar cells are used for operating electronic watches & calculators.
ENERGY FROM THE SEA:
Oceans are large water bodies which covers about 70.8% of the Earth’s total surface area. They are the huge reservoirs of energy which can be obtained for useful purposes in a variety of ways. Following are the most common ways of obtaining the energy from the oceans:
TIDAL ENERGY:
Tides are everyday movement of water level along the coasts. Tides are due to the gravitational pull of the moon on the spinning Earth. This pull varies during the monthly cycle of rotation of the moon around the earth. The level of water may rise upto a few metres in high tides which occur on every new moon day and full moon day.
The energy derived from rising and falling ocean tides is called tidal energy. Tidal energy is harnessed by constructing a dam. A turbine fixed at the opening of the dam converts tidal energy to electricity. However, energy generation requires:

(a) Merits and demerits of tidal energy
The various merits and demerits of tidal energy are :
Merits
- It is an inexhaustible and renewable pollution-free source of energy.
- It is independent of uncertainty of rainfall.
Demerits
- Power generation is intermittent due to variation in tidal range.
- The most difficult problem in the use of tidal power is the barrage construction in areas of high tidal flow and corrosion of barrage.
WAVE ENERGY:
Due to the blowing of wind on the surface of ocean, very fast sea-waves move on its surface. Due to their high speed, sea waves have a lot of kinetic energy in them. The energy of moving sea-waves can be used to generate electricity. This can be done as follows :
(i) One idea is to set up floating generators in the sea. These would move up and down with the seawaves. This movement would drive the generators to produce electricity.
(ii) Another idea is to let the sea-waves move up and down inside large tubes. As the waves move up, the air in the tubes is compressed. This compressed air can then be used to turn a turbine of a generator to produced electricity.

(a) Merits of wave energy
- It is a renewable and pollution free source of energy.
- It does not require large land areas.
Demerits of wave energy
- The power supply is variable in nature due to variability in wave formation.
- Marine life could be affected due to wave energy harnessing structures.
Ocean thermal energy:
The energy available due to the difference in the temperature of water at the surface of the ocean and at a deeper levels is called ocean thermal energy. The drives used to harness ocean thermal energy are called ocean thermal energy conversion power plants. A temperature difference of 20°C between the surface water of ocean and deeper water is needed for operating OTEC power plants. In one type of OTEC power plant, the warm surface water of ocean is used to boil a liquid like ammonia or a chlorofluorocarbon (CFC). The high-pressure vapours of the liquid are then used to run the turbine of a generator and produce electricity. The colder water from the deeper ocean is pumped up to cool the used up vapours & convert them again into a liquid. This process is repeated again and again.
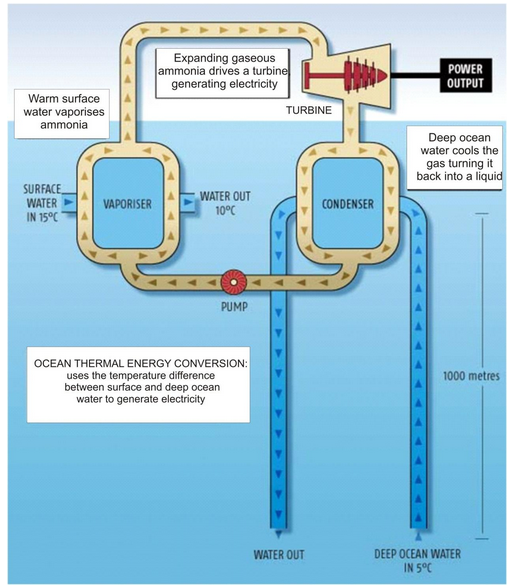
OTEC power plants:
These plants can operate if the temperature differences between the water at the surface and water at depths upto 2 km is 293 K (20°C) or more. The plant undergoes the following cycle of operations.
-
The warm surface water is used to vapourize pressurized ammonia (approx. 8 atm) in an evaporator (i.e., a heat exchanger) through which warm sea water flows.
-
The vapours are then used to turn a turbine of generator to generate electricity.
-
The cold ocean water is transported to the surface and is used to condense ammonia vapour through a condenser.
-
The ammonia condensate (condensed ammonia vapour) is pumped back to the evaporator through a pump.
Merits of OTEC
- The electric power produced is continuous, renewable and pollution free.
- OTEC system does not have daily or seasonal variations in their output as in case with the solar energy devices.
Demerits of OTEC
- OTEC system requires a lot of capital investment.
- The conversion efficiency is low as there is small temperature difference between the surface water and the deep water.
GEOTHERMAL ENERGY:
Geothermal energy is the heat energy of hot rocks present inside the earth. This heat can be used as the source of energy to produce electricity. Geothermal energy is one of the few sources of energy that do not come directly or indirectly from solar energy. The places where very hot rocks occur at same depth below the surface of earth are called ‘hot spots’ and are sources of geothermal energy.
The geothermal energy is harnessed as follows:
(i) The extremely hot rocks present below the surface of earth, heat the underground water and turn it into steam. As more and more steam is formed between the rocks, it gets compressed to high pressures. A hole is drilled into the earth and the hot rocks comes up through the pipe at high pressure. This high-pressure steam run the turbine of a generator to produce electricity.
(ii) Large rocks are present in the underground rocks, which allow steam and hot water to go up. The steam & hot water gushing out of the ground are a kind of natural geyser. This steam is then used to turn turbines and generate electricity and the hot water is used to cook food.

Geo thermal energy
Merits of geothermal energy:
- It is the most versatile and least polluting renewable source of energy.
- It is relatively inexpensive and power generation level is higher as compared to solar energy and wind energy.
Demerits of geothermal energy:
- Though geothermal energy is inexhaustible, a single bore has a limited life span of about 10 years.
- Geothermal hot spots are scattered.
NUCLEAR ENERGY:
The energy obtainable from the nucleus of an atom is called nuclear energy or atomic energy.
Nuclear energy is measured in terms of an electron volt (eV) where 1 ev = 1.6 × 10−19 J.There are two distinct ways of obtaining nuclear energy
NUCLEAR FISSION:
In 1939 two German scientists Otto Fiahn and Friiz Strassman very carefully analysed the products of their experiments on bombardment of uranium with neutrons. One of the product was found to be a barium isotopes emitting β−rays and having a half life of 86 minutes. This was identical to the known characteristics of 138Ba. Another product of this reaction was an isotope of lanthanum 140La, which had a half-life of 40 hours. The uranium atom with Z = 92 and atomic mass number of 235 disintegrates into atoms, whose atomic number is Z = 56 and atomic mass number = 139 (barium) and Z = 57 has atomic mass number = 140 (lanthanum). The mass of the products = 139 + 140 = 279 u [unified atomic mass unit]. This is more than the mass of uranium nucleus and therefore only one of the two products would be produced in a given reaction. The other product would be an atomic species with atomic number of nearly 36 and atomic mass of about 100 u. Nahn and Strassman were indeed able to find active isotopes of Strontium 38Sr, and Yttrium 39Y.
Nuclear fission is the phenomenon of splitting of an unstable nucleus of a heavy atom (like uranium-235, plutonium or thorium) into two medium weight nuclei with the liberation of an enormous amount of energy.
|
|
A nuclear reaction in which the particle which initiates the reaction is also produced during the reaction and it carries on the reaction further and further, is called a nuclear chain reaction. This type of nuclear reaction is called uncontrolled nuclear reaction and is the basis of an atom bomb.
Nuclear chain reaction |
Fission Products:
Fission of uranium produces nuclei that have mass number range from 72[72Zn30] to mass number 158 [158E63]. Nuclei of different mass numbers can be produced by fission of uranium. Fission of 235U92 yields mainly two group of nuclei. One of the group is a light group with atomic mass numbers in the range 85 to 104 u. The second group is a heavy group with mass numbers range from 130 to 149 u. Most commonly occurring products are molybdenum [42Mo] and lanthanum [57La]. There are some "PROMPT" and "DELAYED" categories. In first category prompt, the uranium nucleus splits immediately when a projectile of proper energy strikes. In delayed fission, the projectile enters the nucleus and causes instability which leads to fission of host nucleus, 236U92. The fission of natural uranium and some other isotope take place without any projectile being hit on the same.
Types of Fission Reactions:
All the nuclear fission reactions can be divided into three categories:
(i) Spontaneous fission (ii) Prompt fission and (iii) Delayed fission.
(i) When a nucleus undergoes fission on its own (without being hit by a projectile like neutron), it is called spontaneous fission.
A spontaneous nuclear fission does not need to be initiated. In spontaneous nuclear fission, the natural shaking motion (or oscillations) of the nucleons in a heavy nucleus causes it to break into smaller nuclei. Spontaneous fission keeps on taking place in natural uranium all the time, but at a very-very slow rate.
(ii) When a nucleus splits up into smaller nuclei instantaneously as soon as it is bombarded with a projectile (like neutrons), it is called prompt fission. About 99 percent of the uranium-235 fission caused by slow neutrons is prompt fission. The prompt fission is produced by prompt neutrons which are emitted instantaneously by the splitting nuclei.
(iii) When a projectile like neutron enters a nucleus and causes an instability which leads to the fission of the nucleus after a short while, it is called delayed fission. About 1 percent of the uranium- 235 fission caused by neutrons is delayed fission. The delayed fission is caused by delayed neutrons, which are emitted slowly by the splitting nuclei.
Different amount of energies are required to produce fission from one isotope to the other. There is threshold energy of projectiles below which no fission takes place.
Fission of nuclei is often accompanied by emission of neutrons that cause further fission. The fission of uranium is accompanied by 2 or 3 neutrons per fission. Energies of such neutrons lie in a wide range.
Unit of energy usually used in nuclear reactions is eV or MeV.
1eV = 1.6 x 10−19 J
1 MeV = 1.6 x 10−13 J
(c) Energy Released by Fission Reaction:
The energy released in a fission reaction can be calculated by comparing the masses of the nucleus which has undergone fission together with that of the projectile used to cause fission and the masses of fission products.
e.g. 1 Consider fission of 235U92 , fission takes place when it is bombarded with low energy neutrons (neutrons having an energy of about 0.025 eV). Velocity of such neutrons is about 2,200 ms−1 which is about ten times that of modern jet plane. There are many fission reactions possible. Consider most portable reaction in which 95Mo35 and 139La57 is produced with emission of 2 neutrons
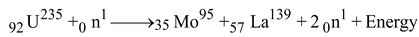
Mass of 92U235 = 235.124 amu
Mass of 1 0n1 =1.009 amu
Initial mass = 236.133 amu …(i)
Mass of 35Mo95 = 94.946 amu
Mass of 57La139 = 138.955 amu
Mass of 2 10n= 2.018 amu
Final mass = 235.919 amu …(ii)
We find that the initial mass is more than final mass. This means that mass defect [(i) - (ii)] i.e.,
236.133 u − 235.919 u = 0.214 amu
We know that 1 amu or 1 u gives 931 MeV energy.
 0.214 x 931 = 199.234 ≈ 199.2 MeV of energy is released.
0.214 x 931 = 199.234 ≈ 199.2 MeV of energy is released.
e.g. 2 An another popular nuclear reaction of 92U235 is,
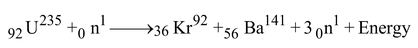
Initial mass = mass of 92U235 + mass of 0n1 = 235.0439 + 1.0087 = 236.0526 amu
Final mass = mass of 36Kr92 + mass of 56Ba141 + 3 x mass of 0n1 …(i)
Final mass = 91.8954 + 140.9177 + 3.0261 = 235.8392 u …(ii)
Difference of mass = 236.0526 amu - 235.8392 u = 0.2134 amu
But 1 amu gives 931 MeV energy.
 Energy produced = 0.213 amu x 931 = 198.7 MeV
Energy produced = 0.213 amu x 931 = 198.7 MeV
e.g. 3 Energy produced from 1 Kg of 92U235 .
We know that 235 g of 92U235 contains 6.023 X 1023 atoms
Number of atoms in 1 Kg of 92U235 = (6.023 x 1023/235) x 1000 atoms
= 25.63 × 1023 atoms
If one atom releases 200 MeV of energy, then energy released by 1Kg of 92U235 =25.36 x 1023 x 200 MeV
= 5.126 x 1023 MeV
= 5.126 x 1023 x (1.6 x 10−13J)
=8.2014 x 1010J = 8.201 x 1010J
This is the energy produced by burning 2,500 tons of coal.
Process of Nuclear Fission:
The process of nuclear fission is explained by the 'Liquid drop model' of the nucleus. The liquid drop model of the nucleus to explain the process of fission was proposed by “Yakov Frenkel, Neils Bohr, and John Wheeler”. According to the liquid drop model of the nucleus, it is said that in just the same way that a drop of water might become unstable if another small drop hits it, the uranium nucleus becomes unstable and breaks up when hit by a neutron. In this model, the uranium nucleus is treated as a drop of a liquid, which is not compressible and has a uniform positive charge. It is imagined that a stretchable skin-like membrane surrounds the drop-like nucleus and holds all the protons and neutrons together inside its body. In the stable stage, the uranium nucleus, like a drop of water is spherical. The nuclear fission of uranium - 235 isotopes using a slow-moving neutron can be explained diagrammatically as follows:
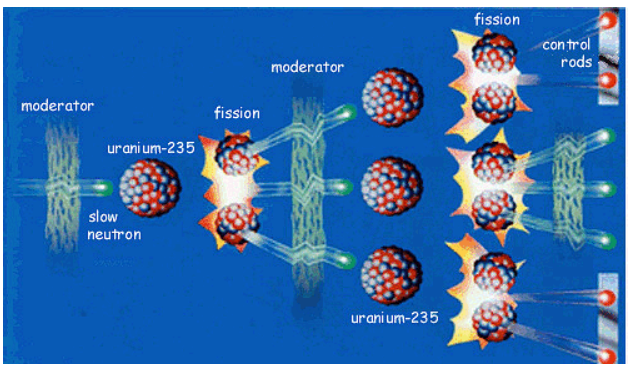
The figure above shows that the uranium nucleus is spherical. In this spherical nucleus, the nuclear particles like protons and neutrons are very close together because of which the nuclear force of attraction and the electrostatic force of repulsion are very delicately balanced. Now, when a slow-moving neutron attacks the uranium- 235 nuclei and enters it, then the delicate balance of force inside the nucleus is disturbed. The energy of the neutron is transferred to the nucleons (protons and neutrons) and by gaining this energy, the nucleons start oscillating more and more. Due to increased oscillations of the nuclear particles, the skin-like outer membrane of the nucleus gets stretched, the nucleus gets elongated and a waist develops in it. When the nucleus gets elongated the distances between nuclear particles (protons and neutrons) increase. This increased inter-particle distance weakens the nuclear force of attraction so that the electrostatic force of repulsion becomes more dominant. Due to the increased repulsion between the protons, a neck develops in the nucleus as shown. The formation of the neck decreases the nuclear force further. And the increased repulsion between protons ultimately leads to the breaking up of the uranium-235 nucleus to form two smaller nuclei of barium-139 and krypton- 94, along with the emission of three neutrons.
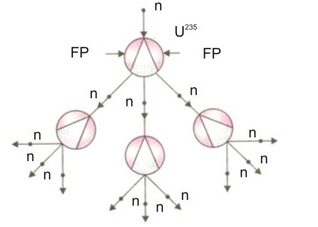
(e) Chain reaction :
A reaction in which the particle which initiates (starts) the reaction is also produced during the reaction to carry on the reaction further and further is called a chain reaction. Once started a chain reaction will go on propagating by itself, until one of the reactant is all used up. The fission of uranium- 235 by means of slow moving neutrons is a chain reaction, because this reaction is started by neutrons and neutrons are also produced during this reaction. The neutrons produced during the fission of a uranium atom initiate the fission of more uranium atoms, and this process goes on, like an unending chain, with the liberation of a large amount of energy at each step. The chain reaction taking place during the fission of uranium- 235 can be represented more clearly with the help of a diagram.
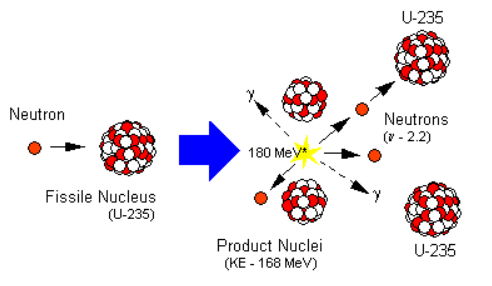
The fact that only 1 neutron is used up in each fission process but 3 neutrons are produced, makes the fission process in uranium-235 a self-sustaining process or self-propagating process called chain reaction. If an average less than 1 neutron had been produced per fission, then the reaction would have died down like a fire in a wet fuel. It should be noted that a chain reaction involves a large number of uranium atoms. So, a chain reaction cannot occur in a very small lump of uranium- 235 isotopes, but it can take place in a sufficiently large mass of uranium-235 isotopes. The minimum mass of uranium- 235 atoms or any other fissionable material which can support a chain reaction is called critical mass of that material.
NUCLEAR FUSION:
Under special conditions, it is possible that nuclei of light elements can combine to form a nucleus of higher atomic number. For example, two deuterons can join together to form a 3He nucleus plus a neutron. The deuteron (D) is the heavy hydrogen nucleus (2H1) consisting of one proton and one neutron. In simple language, two heavy hydrogen nuclei can combine to form a nucleus of helium (3He). This process, in which nuclei of low atomic numbers combine to form a heavier atomic nucleus, is known as nuclear fusion. The fusion reaction is also accompained by release of energy, like the fission phenomena. Fusion was discovered nearly 70 years ago in 1930. The typical fusion reaction mentioned above can represented by the following equation:
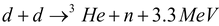 …..(i)
…..(i)
Here, d represents the deuteron. Reaction is also possible:
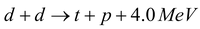 …..(ii)
…..(ii)
Here, t is a triton (proton + two neutrons) or a heavy hydrogen nucleus (3H1) with two extra neutrons. This means that two deuterons can fuse to form either 3He + n, or triton + a proton. Another type of important fusion reaction involves conversion between neutron and protons. The simplest among these is the synthesis of deuteron from hydrogen by the following reaction:
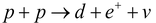 …..(iii)
…..(iii)
In this reaction one of the two protons is converted into a deuteron with simultaneous creation of a positron e` (identical to an electron except that it has a positive charge) and a neutrino v. The neutrino is a neutral particle whose mass is negligible and therefore has enormous penetrating power.
Fusion has been observed in a variety of low mass nuclides.
The fusion reactions can be produced in the laboratory by accelerating low atomic number nuclei in a particle accelerator. The high - energy projectile is made to strike on a target made up of material whose nuclei can fuse with the projectile nucleus.
The kinetic energy of nuclei like deuteron can be sufficiently high at temperatures of 106 K for the nuclei to fuse. The fusing nuclei had to overcome the potential energy due to very high electrostatic repulsion at extremely small distances. However, If the kinetic energy due to thermal motion is increased sufficiently at elevated temperatures of millions of Kelwin, this repulsion can be overcome. At such high temperatures, all the atoms are completely ionised. There is a mixture of electrons and nuclei, which are moving at high speeds. This type of material consisting of moving charged particles with equal number of negative and positive charges is termed as a plasma. The nuclear fusion under such conditions is termd as thermo - nuclear reaction.
In 1939, Hans Sethe proposed that the enormous amount of energy being produced in the sun is due to thermo-nuclear reactions taking place there. This hypothesis has now become universally accepted.
Source of Sun’s Energy:
The sun is a huge mass of hydrogen gas and the temperature in it is extremely high. In the sun the small deuterium atoms (isotope of H atom) collide and fuse together to form bigger atoms of helium. Each time an atom of helium is formed, tremendous energy is released in the form of heat and light. It is this energy which makes the sun shine. The main reaction taking place in sun is -
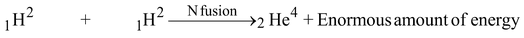
Deuteron Deuteron Helium
(heavy hydrogen) (nucleus)
Sun also contains two other isotopes of H: ordinary H, 1H1 (called protium) and very heavy hydrogen 3H1 (called tritium).
These two also fuse with deuterium to form helium nucleus and release energy. Thus two more reaction taking place in sun are
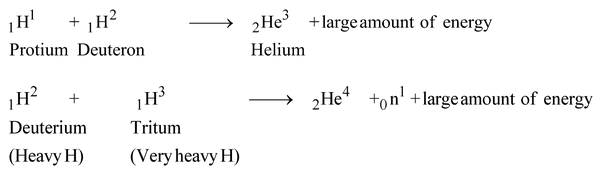
Nuclear fusion reactions of various isotopes of hydrogen to form helium are going on inside the sun that produces such a great amount of energy which is radiated by the sun. Because of this energy; sun emits radiations (light) of different wavelengths of all the sun's radiation. It is the infra-red radiations which carry heat energy and hence heat up the earth.
Hydrogen Bomb:
The nuclear fusion is the basic principle involved in the preparation of hydrogen bomb. A hydrogen bomb is actually an uncontrolled nuclear fusion process.
A hydrogen bomb consists of an arrangement of nuclear fission in the centre. it is surrounded by a mixture of deuteron 2H1 and lithium - 6 isotope (3Li6).
The nuclear fission provides heat and neutrons. The neutrons are used in converting lithium isotope into tritium 3H1 and the heat liberated is required for the fusion between 1H2 and 1H3 to start. The fusion reactions are then accompanied by the liberation of a large amount of energy.
Note: The nuclear fusion reaction occur at very high temperature, so they are also known as thermonuclear reactions.
The reactions are:

(Note: - Tritium has to be prepared within the hydrogen bomb because it is not stable)
DIFFERENCE BETWEEN NUCLEAR FISSION AND NUCLEAR FUSION:
|
Nuclear fission |
Nuclear fusion |
|
1. It is confined to heavy nuclei only. |
1. It is confined to lighter nuclei |
|
2. A heavy nucleus splits into two lighter nuclei . |
2. Two lighter nuclei fuse to form a heavy nucleus. |
|
3. It is a chain reaction . |
3. It is not a chain reaction. |
|
4. Temperature required for the reaction is not high. |
4. It is a thermo nuclear reaction i.e. it requires high temperature. |
|
5. Fission reaction can be controlled . |
5. Fusion reaction is very difficult to control. |
|
6. Large amount of energy is released. |
6. Energy released is far more than that released in a fissoin reaction. |
|
7. Large number of radioactive products are obtained i.e. large amount of nuclear waste is left |
7. No nuclear waste is left |