
Dihydrogen H2 Molecules
Hydrogen of Class 11
Dihydrogen H2 Molecules
(i) The term dihydrogen is used for the mixture of diatomic molecules such as H2,D2 and HD containing H and D atoms with respect to their natural abundance.
(ii) For diatomic molecules containing only protium atoms (H2) the term used is diprotium.
(iii) For diatomic molecules containing only deuterium atoms (D2), the correct term is dideuterium.
Occurrence of Hydrogen
Hydrogen is most abundant element in the universe. About half the mass of sun and stars is accounted for by hydrogen. Jupiter and Saturn mostly consist of hydrogen. On earth, it occurs in free state in small traces in volcanic and natural gases. However in combined state it is widely distributed as water.
Preparation of Dihydrogen
Some of the important methods of preparation of dihydrogen are given below:
1. By the action of water on metals.
Various metals react with water at different temperatures to displace hydrogen from it. For example,
(a) Very active metals like Na, K, Ca react at room temperature to form H2 along with corresponding hydroxides.
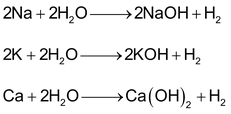
Reaction with these metals is highly vigorous and H2 liberated immediately catches fire which can cause accident. In order to slow down the reaction, amalgam of these metals with mercury are used.
(b) Metals like Mg, Al, Zn, etc, react with water at boiling temperature to form H2 and corresponding oxides.
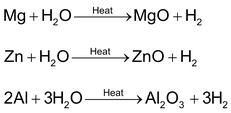
(c) Metals like Fe, Sn, etc, react with steam at high temperature.
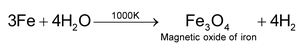
The reaction between iron and steam is used for the large scale preparation of dihydrogen by Lane’s Process.
2. By the action of acids on metals
Metals lying above hydrogen in the activity series displace hydrogen by reaction with dilute HCl or dilute H2SO4.
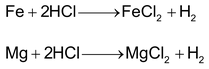
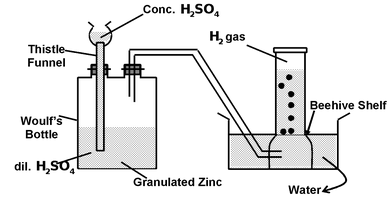
In the laboratory, dihydrogen is prepared by the treatment of zinc granules with dil.H2SO4.
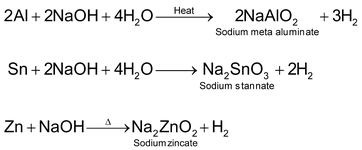
3. By the action of strong alkalies on metals
Metals like Zn, Sn or Al react with strong alkalies such as NaOH or KOH liberating H2 gas
4. Electrolysis of water
Dihydrogen of high degree of purity is prepared by the electrolysis of water in the presence of small amount of acid or base.

Laboratory Preparation of Dihydrogen
In the laboratory, dihydrogen is prepared by the action of diluted sulphuric acid on granulated zinc according to the arrangement shown in the figure

Commercial Preparation of Dihydrogen
From steam and water gas- Lane’s process
Dihydrogen is manufactured by passing alternate currents of steam and water gas over red hot iron at about 1025-1075 K. The method, in fact, consists of two stages.
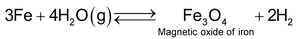
(a) Oxidation stage
It involves passing of super heated steam over iron fillings heated to about 1025—1075K. Iron gets oxidized to Fe3O4 and in turn it reduces water to dihydrogen gas.
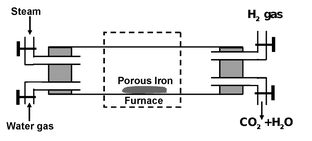
(b) Reduction stage.
When whole of iron is oxidized to Fe3O4, the supply of steam is cut off and stream of water gas (CO + H2) is passed so as to cause reduction of Fe3O4 to back to iron.
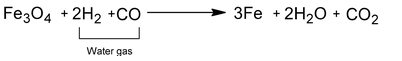
The oxidation and reducing periods are alternatively carries out using two or more furnaces.
Bosch’s Process
The maximum quantity of commercial dihydrogen is prepared by this method. It involves the following steps:
(a) Preparation of water gas
It is prepared by the action of steam with red hot coke
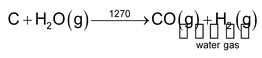
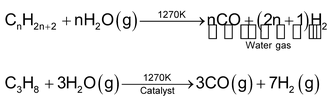
Water gas also be obtained by action of steam on hydrocarbons in the presence of catalyst. This process is also called steal reforming of hydrocarbons
(b) Separation of Hydrogen
The water gas formed by any of the above methods, is mixed with steam and is passed over heated Fe2O3 and Cr2O3 at 770K when CO is oxidized to CO2. Gaseous mixture of CO2 and H2 is then bubbled into cold water under pressure when CO2 dissolves leaving behind dihydorgen gas which escape out.
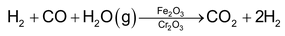
This reaction is called water gas-shift reaction.
Electrolysis of Water
This process involves the electrolysis of acidified or alkalined water. In general, the electrolysis is carried out using iron as cathode, nickel coated iron as anode and 15-20% NaOH solution as electrolyte. The reaction taking places are
Cathode: Reduction of water occurs

Anode: Oxidation of OH occurs
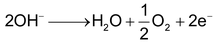
Properties of Dihydrogen
Physical Properties
1. It is colourless, tasteless, odourless gas.
2. It has extremely low solubility in water. Since its molecules are non-polar.
3. Its density is approximately 1/4th that of air. So lightest substance known.
4. It can be liquefied at high pressures and very low temperature.
5. It is highly combustible and should be handled with care.
Chemical Properties
Because of high dissociation energy (435.9 kJ mol-1) dihydrogen is not very reactive. However, it forms compounds with large number of elements under appropriate experimental conditions. Some of the chemical properties are:
1. Neutral character
It is neutral to litmus.
2. Combustibility
It is highly combustible gas. It burns in air with a pale blue flame to form water.
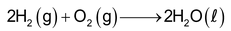
3. Reaction with metals.
It reacts with metals like Na, Ca, Li, etc, to form respective hydrides. In such reactions it acquires the oxidation state of -1 and acts as an oxidizing agent.

In case of metals like Pt, Pd, Ni, etc, hydrogen forms interstitial hydrides in which hydrogen atoms occupy the interstitial in the metallic crystals. This property is refereed to as occlusion. The occluded hydrogen can be liberated from the metals by strong heating.
4. Reaction with non-metals
The chemical reaction of dihydrogen non-metals have been summarized as follows:
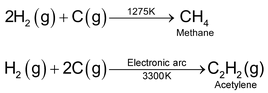
Carbon :
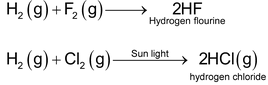
Halogen :
Dioxygen :
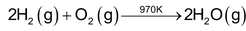
Dinitrogen :
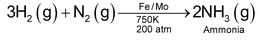
5. Reaction with metal oxide
Reduces such as iron and metal less active than iron.
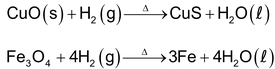
6. Reaction with carbon monoxide
Dihydrogen reacts with carbon monoxide at 700 K in the presence of a catalyst Zn/Cr2O3 to produce methanol.
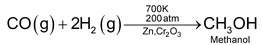
7. Reaction with unsaturated hydrocarbons
Unsaturated hydrocarbons like ethene (CH2 = CH2) and ethyne (CH ≡ CH) react with dihydrogen to form saturated hydrocarbons.
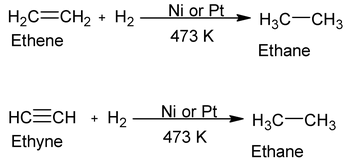
8. Hydrogenation of Vegetable oils.
When dihydrogen (under pressure) is passed through edible vegetable oils such as ground – nut or cotton seed oil at about 473 K in presence of finely nickel as a catalyst, they undergo hardening and change into edible fats known as Vanaspati Ghee.
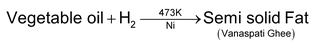
The above process is known as hydrogenation or hardening of oils.
Uses of Dihydrogen
Dihydrogen is used in various industries as listed below:
(i) In the preparation of ammonia which is a starting material for the manufacture of various fertilizers such as urea, ammonium sulphate, calcium ammonium nitrate, etc.
(ii) In the hydrogenation of vegetable oils.
(iii) In the manufacture of synthetic petrol.
(iv) In the oxy-hydrogen torch for welding, if the temperature around 2500°C is desired.
(v) In the atomic hydrogen torch for welding, if the temperature around 4000°C is desired.
(vi) Liquid hydrogen mixed with liquid oxygen is used as rocket fuel in space programmes.
