
Pigment Systems or Photosystems
Photosynthesis In Higher Plants of Class 11
Photosystems
Photosynthetic units occur in the form of two distinct groups called photosystems or pigment systems. Green plants and cyanobacteria possess two photosystems, I and II. But bacteria possess only one photosystem.
Photosystem I(PS I). It is a photosynthetic pigment system along with some electron carriers that is located on both the non appressed part of grana thylakoids as well as stroma thylakoids. PS I has more of chlorophyll a. Chlorophyll b and carotenoids are comparatively less. Its photocentre has a special chlorophyll a molecule called P700 (P703). It is surrounded by other chlorophyll a molecules, followed by chlorophyll b and carotenoids. Photosystem I has FeS, ferredoxin, plastoquinone, cytochrome complex and plastocyanin. It takes part in both cyclic and non-cyclic photophosphorylation. PSI can carry on cyclic photophosphorylation independently. Normally it drives an electron from photosystem II to NADP+.
Photosystem II (PS II). It is a photosynthetic pigment system along with some electron carriers that is located in the appressed part of the grana thylakoids. PS II has chlorophyll a, b and carotenoids. Chl a and chl b contents are almost equal. Carotenoid content is higher as compared to that of PS I. The photocentre is a special chlorophyll a molecule called P680 (P683). It is surrounded by other chlorophyll a molecules, chlorophyll b molecules. PS II also contains Mn2+ Cl–, quencher molecule Q, plastoquinone (PQ), cytochrome complex and plastocyanin. It picks up electron released during photolysis of water. The same is extruded on absorption of light energy. As the extruded electron passes over cytochrome complex, sufficient energy is released to take part in the synthesis of ATP from ADP and inorganic phosphate. This photophosphorylation is non-cyclic. PS II can operate only in conjunction with PS I.

Table : comparison between Photosystem I (PS I) and Photosystem II (PS II)
Absorption Spectrum
Graphic representation of degree and portions of light wavelengths absorbed by a substance is called absorption spectrum. Recording of wavelengths of light absorbed by a substance is made with the help of an instrument called spectrophotometer. In an absorption spectrum the height of curve at a given wavelength gives the relative absorbance at that wavelength. Absorption spectra of chlorophylls a and b show that the two pigments absorb maximum in blue-violet and red parts of light. It has been found that chlorophyll a has several forms which show maximum absorbance in red region at different wavelengths like 660–670, 670–680, 690–700 nm etc.
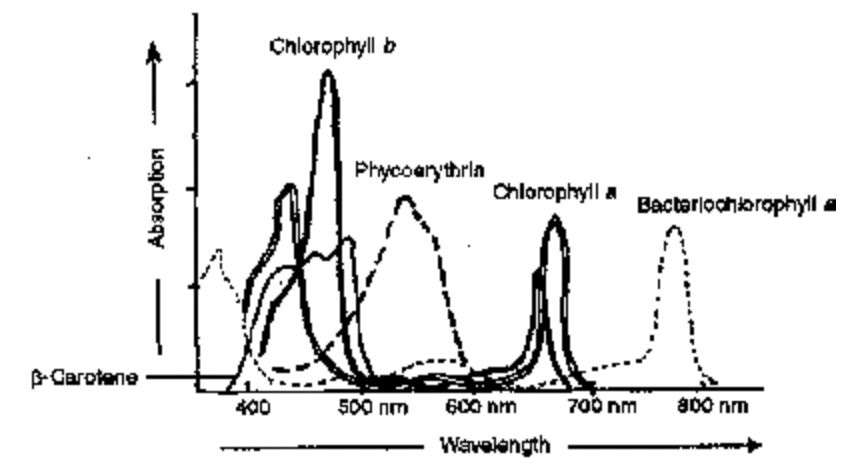
Fig. Absorption spectra of some photosynthetic pigments
Action Spectrum
It is a graph that depicts the effectiveness of light of various wavelengths in performing a particular function. The first action spectrum of photosynthesis was studied by T.W. Engelmann (1882) using green alga Spirogyra and aerobic bacteria. Engelmann employed a prism to break sunlight into rainbow of different colours that fell over the aquatic alga. Oxygen seeking bacteria were found to accumulate around regions of alga according to the amount of photosynthesis performed and oxygen evolved. They were regions which received blue and red lights.
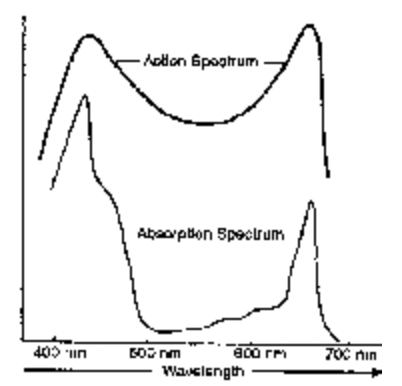
Fig. Action spectrum of photosynthesis and absorption spectrum for leaf extract
Action spectrum of light dependent response should closely resemble the absorption spectrum of a pigment or pigment complex that absorbs the effective light. A comparison of action spectrum of photosynthesis with absorption spectra of various photosynthetic pigments indicates that both chlorophyll a and chlorophyll b takes part in photosynthesis in blue and red parts of spectrum. In the mid part of spectrum photosynthesis is comparatively less. Here light is absorbed by carotenoids.
Fluorescence
It is almost instant re-radiation of light energy by a substance after having absorbed the same. Fluorescence stops immediately after the withdrawal or source of illumination. All types of photosynthetic pigments are fluorescent. However, most of the fluorescence produced by green parts are due to chlorophyll a as all other pigments hand over absorbed energy to it through resonance. On absorption of light energy, a fluorescent substance gets excited. In the excited state, an outer electron moves to a higher orbital. It is called excited singlet state. Liberation of unutilised absorbed energy is called fluorescence. Chlorophyll emits red fluorescence though it absorbs both blue and red wavelengths of light. It has two peaks, one in the red region below 700 nm and the other in the far-red above 720 nm. ‘The out burst of fluorescence on initial few seconds of illumination is called as Kutusky effect’.
Phosphorescence
Fluorescence is almost immediate. If whole of absorbed energy is not lost in fluorescence, the excited molecule loses a small amount of energy and comes to lie in triplet excited state. Afterwards it loses the remaining energy and comes to ground state. The delayed re-radiation of absorbed energy is called phosphorescence.
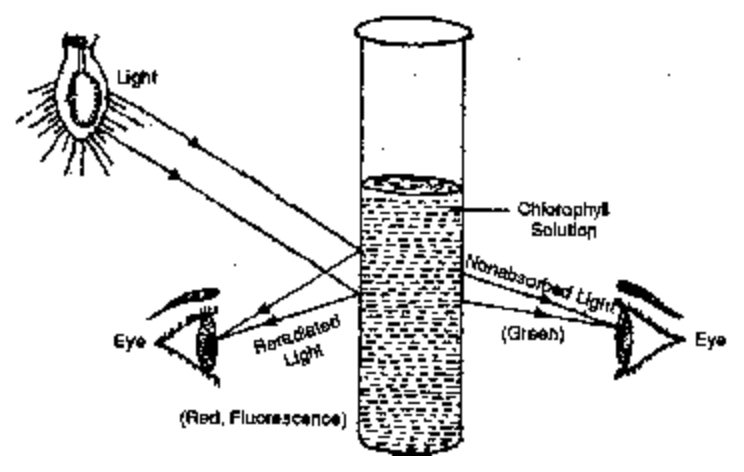
Fig. Chlorophyll colour in nonabsorbed transmitted and re-radiated light
