How Haloform reaction proceed ?
Haloform Reaction
The general reaction is represented as

The CH3 carbon is lost as CHX3 and the remaining part exists as acid salt, which can be acidified to liberate free acid.
The structural feature essential in the compound to show haloform reaction is that any of the following moieties should be present in the molecule attached to some electron−withdrawing group or electron donating group by +I only.


or any other grouping that can be converted to any of the above moieties.
The mechanism of the reaction can be outlined as:
The reaction has 3 important steps. Step I is the oxidation, caused by mild oxidizing agent (hypo halite ion). The second step is base−promoted halogenation and the third step is cleavage of C–C bond.
Haloform Reaction Mechanism

Some of the compounds which responds positively to iodoform test are
CH3CH2OH (only primary alcohol)
 ,
,  ,
,  (secondary alcohols)
(secondary alcohols)
 (only aldehyde) ,
(only aldehyde) , ,
,  ,
,  (ketones)
(ketones)
 ,
, ,
,  ,
,  (after heating with OH–)
(after heating with OH–)
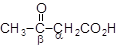 (after heating)
(after heating)
The compounds that respond negatively to iodoform test are
 ,
,  ,
,  ,
,  ,
, 
 ,
, 
The chemical properties of an alcohol, ROH, are determined by its functional group, –OH, the hydroxyl group. Reactions of an alcohol can involve the breaking of either of two bonds: the C-- OH bond, with removal of the –OH group; or the O--H bond, with removal of –H. Either kind of reaction can involve substitution, in which a group replaces the –OH or –H, or elimination, in which a double bond is formed. Differences in the structure of R cause differences in reactivity, and in a few cases even profoundly alter the course of the reaction. We shall see what some of these effects of structure on reactivity are, and how they can be accounted for. Check out Chemistry Formulas and NCERT Solutions for class 11 Chemistry prepared by Physics Wallah.
Recent Concepts
- How to calculate equivalent weight ?
- What is equivalent weight of oxalic acid
- Atomic weight of elements
- What is Limiting Reagents ?
- Molarity
- Define back bonding in BF3
- Heisenberg's Uncertainty Principle
- What are Inert pair effects
- Quantum Numbers
- What is the boron trifluoride formula?
- What is the Structure of orthoboric acid ?
- What is the Structure of Borax ?
- What is the structure of diboranes ?
- LiAlH4 Reaction and Mechanism
- Grignard reagent and its application in organic reactions
- How Haloform reaction proceed ?
- What is Oxirane ?
- Acidity of Phenols
- Explain the mechanism of Bromination of Phenol
- what is a chemical reaction?









