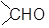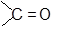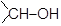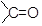LiAlH4 Reaction and Mechanism
LiAlH4 Mechanism
The commonly used reagent for the reduction of aldehydes and ketones are LiAlH4 or NaBH4. The reaction proceeds by successive transfer of hydride ions (H−) from boron or aluminium to four different carbonyl carbons. As all four of the hydrides are transferred, there are distinct reducing agent used in every step of reduction. In the first step, the reducing agent is AlH4− (I), while in the second step, the reducing agent is RCH2OAlH3− (II).
In the same manner, species [RCH2O]2AlH2− (species III) and [RCH2O]3AlH− (species IV) will be obtained. Reduction with NaBH4 is usually done in water or in alcohol which are used as the solvent. The solvent destroys the intermediate alkoxide ion and yields alcohol. If water or an alcohol is not used as the solvent, aqueous acid can be added after the reduction to convert the alkoxide to alcohol.
Lialh4 Mechanism Steps
The lialh4 mechanism for reduction with lithium aluminium hydride is very similar to that of NaBH4. As LiAlH4 violently reacts with water and other polar protic solvents to give molecular hydrogen, reduction with LiAlH4 are done in aprotic solvents like anhydrous ether. Ethyl acetate is added to decompose the aluminium complex. But reduction with NaBH4 can be carried out in water or ethanol solution.Check out Chemistry Formulas and NCERT Solutions for class 11 Chemistry prepared by Physics Wallah.
Mechanism of reduction by LiAlH4:
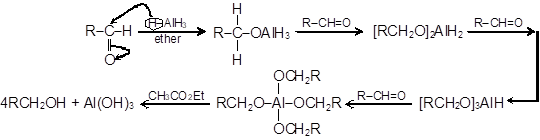
Aldehydes are reduced to primary alcohols by both LiAlH4 and NaBH4 while these reducing agents reduce ketones to secondary alcohols.
(i)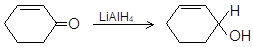
(ii) 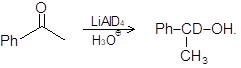
(iii) 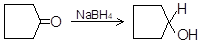
(iv) 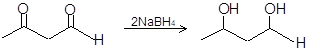
LiAlH4 can reduce all functional groups of column ‘A’ into Column ‘B’. Generally, it is unable to reduce double bond, which are in conjugation with 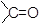 group the double bond is reduced when a phenyl group is attached to the β−carbon.
group the double bond is reduced when a phenyl group is attached to the β−carbon.
Example of LiAlH4
C6H5−CH=CH−CHO 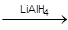 C6H5CH2CH2CH2– OH
C6H5CH2CH2CH2– OH
|
“A” |
“B” |
|
|
−CH2−OH |
|
|
|
|
−COOH |
−CH2−OH |
|
−COOR |
−CH2−OH + ROH |
|
−COCl |
−CH2−OH |
|
(RCO)2O |
RCH2−OH |
|
lactone |
diol |
|
epoxide |
alcohol |
can reduce all the above groups of column ‘A’ into groups of column ‘B’ except, −COOH and −COOR. NaBH4 is able to reduce the double bonds, which are in conjugation with group.
Recent Concepts
- How to calculate equivalent weight ?
- What is equivalent weight of oxalic acid
- Atomic weight of elements
- What is Limiting Reagents ?
- Molarity
- Define back bonding in BF3
- Heisenberg's Uncertainty Principle
- What are Inert pair effects
- Quantum Numbers
- What is the boron trifluoride formula?
- What is the Structure of orthoboric acid ?
- What is the Structure of Borax ?
- What is the structure of diboranes ?
- LiAlH4 Reaction and Mechanism
- Grignard reagent and its application in organic reactions
- How Haloform reaction proceed ?
- What is Oxirane ?
- Acidity of Phenols
- Explain the mechanism of Bromination of Phenol
- what is a chemical reaction?