What is the structure of diboranes ?
structure of diboranes ?
The bonding and structure of the diboranes are of great interest. They are different from all other hydrides. There are not enough valency electrons to form conventional two−electron bonds between all of the adjacent pairs of atoms and so these compounds are term as electron deficient.
In diborane there are 12 valency electrons, three from each B atom and six from the H atoms. Check out Chemistry Formulas and NCERT Solutions for class 11 Chemistry prepared by Physics Wallah.
Electron diffraction results indicate the structure shown in given figure.
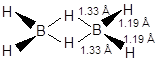
The two bridging H atoms are in a plane perpendicular to the rest of the molecule and prevent rotation between the two B atoms. Specific heat measurements confirm that rotation is hindered. Four of the H atoms are in a different environment from the other two. This is confirmed that diborane cannot be methylated beyond Me4B2H2 without breaking the molecule into BMe3.
The terminal B−H distances are the same as the bond lengths measured in non-electron−deficient compounds. These are assumed to be normal covalent bonds, with two electrons shared between two atoms. We can describe these bonds as two−centre−two−electron bonds (2c−2e), with two electrons shared between two atoms.
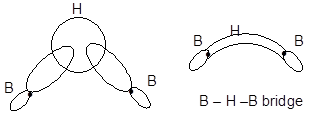
Thus, the electron deficiency must be associated with the bridge groups. The nature of the bonds in the hydrogen bridges is now well established. Obviously, they are abnormal bonds as the two bridges involve only one electron from each boron atom and one from each hydrogen atom, making a total of four electrons. An sp3 hybrid orbital from each boron atom overlaps with the 1s orbital of the hydrogen. This gives a delocalised molecular orbital covering all three nuclei, containing one pair of electrons and making up one of the bridges. This is a three−centre−two−electron bond (3c−2e). A second three−centre bond is also formed.
It contains two 3−centre 2−electron banana bonds (B … H … B). Two electrons from two H atom and two from two Boron atoms consist of four electrons. An overlap of sp3 hybrid orbital of B and 1s hydrogen orbital gives the delocalised molecular orbitals of a B … H … B bridge.
Diboranes
None of the group III elements react directly with hydrogen, but several interesting hydrides are known. The boron hydrides are sometimes called diboranes by analogy with the alkanes (hydrocarbons). Almost 20 diboranes have been reported and 11 are well characterized.
Diboranes fall into two series:
-
BnH(n + 4) (called nido−diboranes).
-
A less stable series BnH(n + 6) (called arachno−diboranes).
Boron hydrides differ from carbon hydrides (i.e. hydrocarbons) that they don’t occur in nature because of their great affinity for H2O and O2. The compound BH3 which would be analogous to BF3 does not exist because H−atom lacks the electrons that would be needed to participate in B−H π−bonding and compensate for the incomplete octet of B. BH3 can exist only in complex formation with donors such as BH3.CO, BH3.N(CH3)3. Molecules of BH3 apparently dimerize spontaneously when they come in contact with each other to form diborane, B2H6.
Recent Concepts
- How to calculate equivalent weight ?
- What is equivalent weight of oxalic acid
- Atomic weight of elements
- What is Limiting Reagents ?
- Molarity
- Define back bonding in BF3
- Heisenberg's Uncertainty Principle
- What are Inert pair effects
- Quantum Numbers
- What is the boron trifluoride formula?
- What is the Structure of orthoboric acid ?
- What is the Structure of Borax ?
- What is the structure of diboranes ?
- LiAlH4 Reaction and Mechanism
- Grignard reagent and its application in organic reactions
- How Haloform reaction proceed ?
- What is Oxirane ?
- Acidity of Phenols
- Explain the mechanism of Bromination of Phenol
- what is a chemical reaction?









