Cannizzaro Reaction
About Cannizzaro Reaction
Those aldehydes (aliphatic or aromatic), which do not have a-hydrogen atom on treatment with strong base undergoes a reaction involving its 2 moles, one getting oxidised to yield acid salt and the other getting reduced to primary alcohol. The important condition is that there should not be a good leaving group attached to the carbonyl group.
.png)
Reaction Mechanism of Cannizzaro Reaction
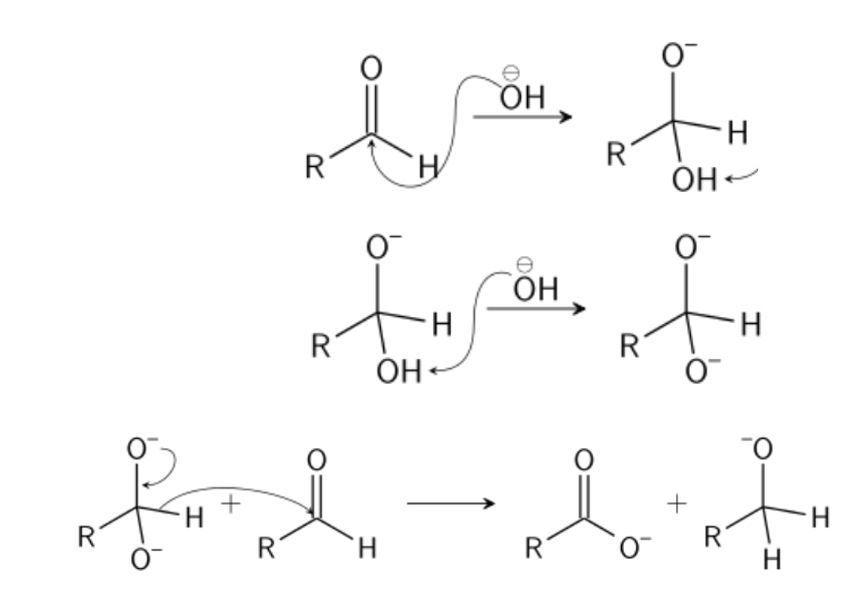
This involves hydride transfer from an aldehyde molecule lacking an α-H atom e.g., HCHO, R3CCHO, ArCHO, to a second molecule of either the same aldehyde (disproportionation) or sometimes to a molecule of different aldehyde (crossed Cannizzaro). The reaction requires the presence of strong bases.
The step by step mechanism of Cannizzaro Reaction are
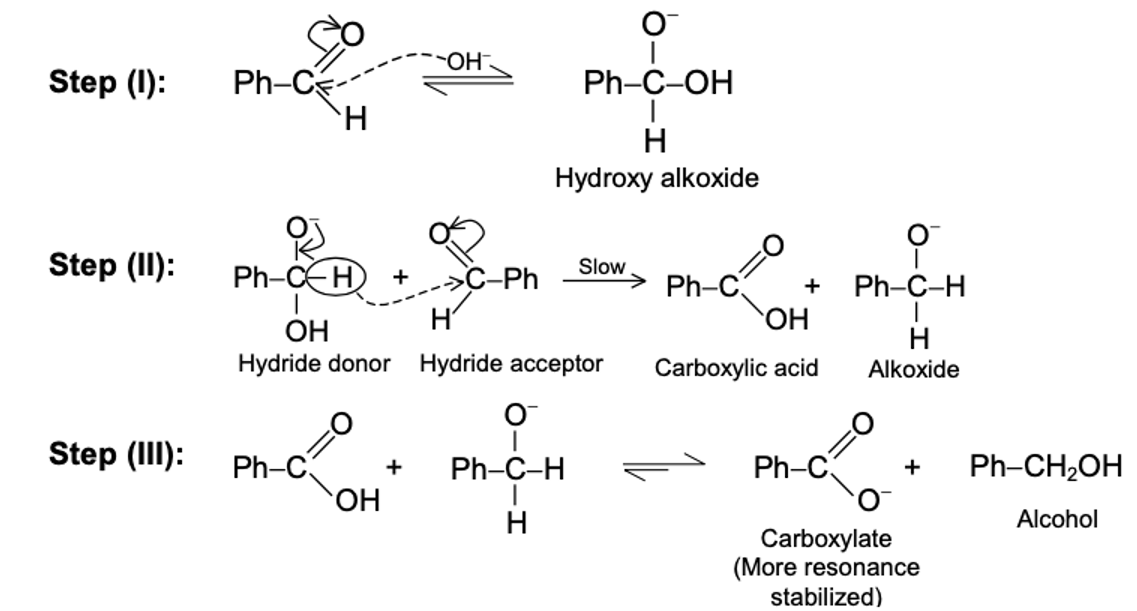
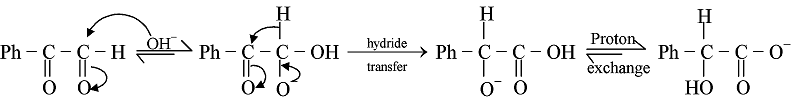
Mechanism of Cannizzaro Reaction at High concentrantion of NaOH
Rapid and reversible addition of -OH to PhCHO yields the hydride donor species, this is followed by slow, rate limiting hydride transfer to the carbonyl carbon atom of a second molecule of PhCHO, and the reaction is completed by rapid proton exchange to yield the more stable pair of carboxylate and alcohol. Mutual oxidation/reduction of two molecules of aldehyde has thus taken place to yield one molecule each of the corresponding carboxylate anion and of the primary alcohol.
Example of Cannizzaro Reaction
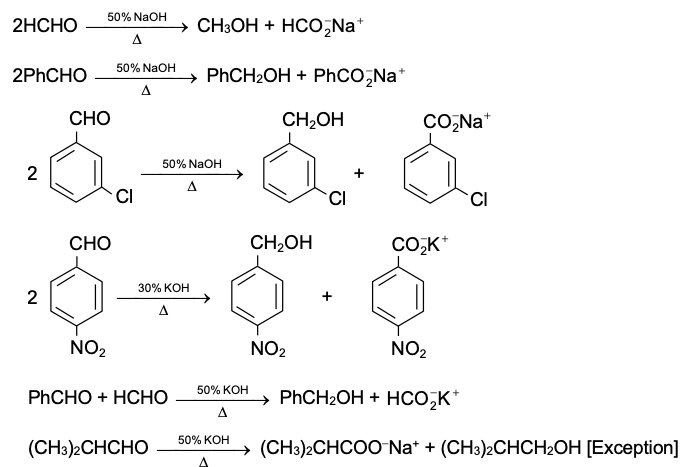
Crossed Cannizzaro reaction
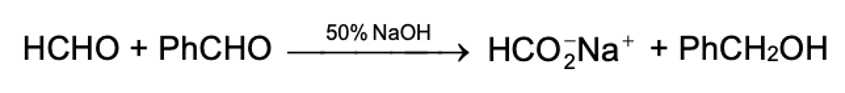
The Cannizzaro reaction between two dissimilar aldehyde moelcuels (crossed Cannizzaro reaction) may yield all possible products. However, when one of the aldehydes is formaldehyde the formate ion and the alcohol corresponding to the other aldehyde are formed exclusively. The exclusive formation of the formate ion is due to the high reactivity of formaldehyde which preferentially adds a hydroxide ion before losing one of its two hydrogens as hydride ion to the less reactive aldehyde.
Intramolecular Cannizzaro
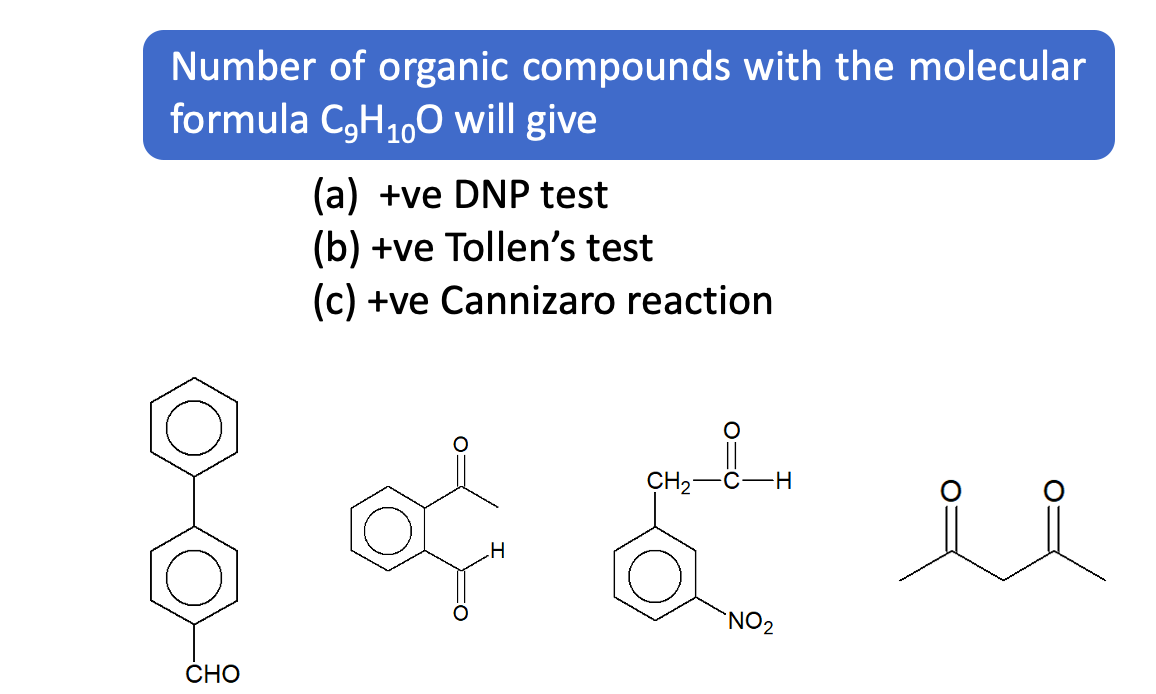
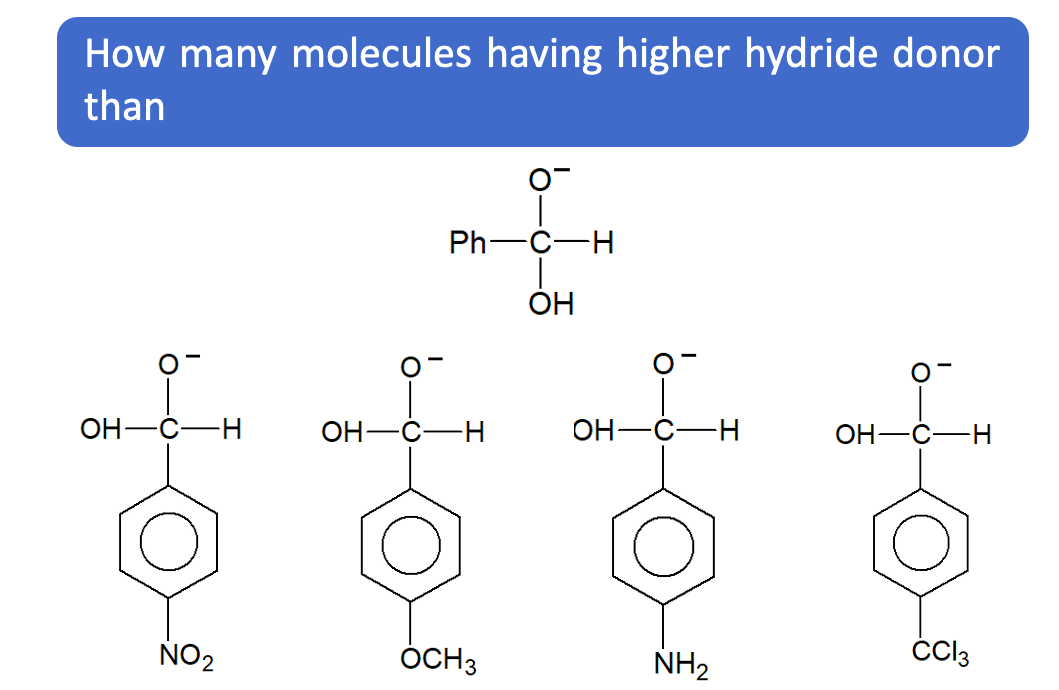
Questions based on Cannizzaro Reaction
List of Name Reaction of Organic Chemistry consist of detail Reaction Mechanism of all name reactions of Organic Chemistry.
Recent Concepts
- Aldol condensation
- Arndt−Ester synthesis
- Baeyer−Villiger Oxidation
- Benzoin Condensation
- Beckmann Rearrangement
- Cannizzaro Reaction
- Clemmensen Reduction
- Claisen condensation
- Etard’s Reaction
- Friedel-Crafts alkylation
- Friedel Crafts Acylation
- Fries Rearrangement
- Gattermann-Koch Reaction
- Grignard Reagent
- Hell-Volhard-Zelinsky Reaction
- Hunsdieker reaction
- Hoffmann Bromamide Degradation
- Jones reagent
- Kolbes Reaction
- Knoevenagel Reaction









