Fries Rearrangement
About Fries Rearrangement
Phenylacetate undergoes the Fries rearrangement with AlCl3 to form ortho and para hydroxy acetophenone. The ortho isomer is separated from the mixture by its volatility with steam.The phenyl ester formed from the reaction of phenol and acid anhydride undergoes rearrangement when treated with anhydrous AlCl3 to yield ortho and para-hydroxyketones. The o-isomer is separated from the mixture by steam-distillation.Generally low temperature (60°C or less) favours the formation of the p-isomer whereas high temperature (above 160°C) favours the o-isomer.
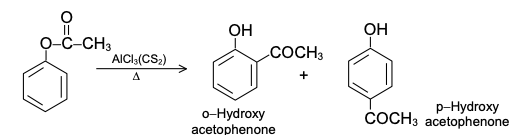
The ortho isomer has higher vapour pressure because of chelation, O–H---O = C and is steam volatile. In the para isomer, there is intermolecular H-bonding with H2O. The para isomer (rate-controlled product) is the exclusive product at 25°C because it has a lower ΔH and is formed more rapidly. Its formation is reversible, unlike that of the ortho isomer which is stabilized by chelation. Although it has a higher ΔH, the ortho isomer (equilibrium-controlled product) is the chief product at 165°C because it is more stable
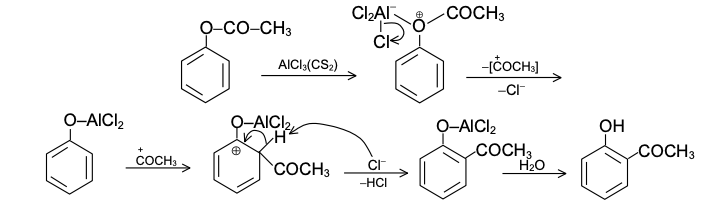
Reaction Mechanism of Fries Rearrangement
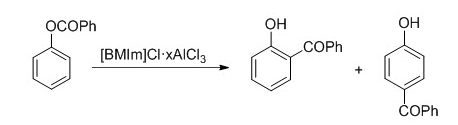
Examples of Fries Rearrangement
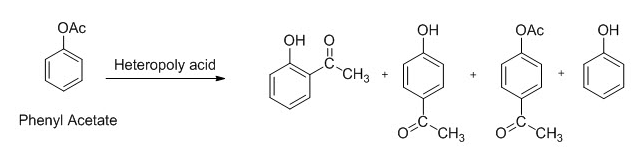
List of Name Reaction of Organic Chemistry consist of detail Reaction Mechanism of all name reactions of Organic Chemistry.
Recent Concepts
- Aldol condensation
- Arndt−Ester synthesis
- Baeyer−Villiger Oxidation
- Benzoin Condensation
- Beckmann Rearrangement
- Cannizzaro Reaction
- Clemmensen Reduction
- Claisen condensation
- Etard’s Reaction
- Friedel-Crafts alkylation
- Friedel Crafts Acylation
- Fries Rearrangement
- Gattermann-Koch Reaction
- Grignard Reagent
- Hell-Volhard-Zelinsky Reaction
- Hunsdieker reaction
- Hoffmann Bromamide Degradation
- Jones reagent
- Kolbes Reaction
- Knoevenagel Reaction









