Wolf Kishner Reduction
Introducation to Wolf Kishner Reduction
About Wolf Kishner Reduction
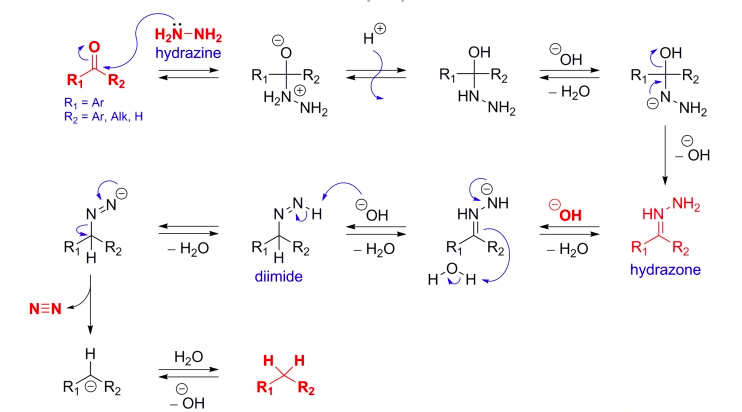
This is a selective reduction it can reduce only carbonyl compounds unable to reduce the OH group. When a ketone or an aldehyde is heated in a basic solution of hydrazine, the carbonyl group is converted to a methylene group this process is called Deoxygenation because an oxygen is removed from the reactant. The reaction is known as the Wolf-Kishner Reduction.
.png)
Initially, the ketone reacts with hydrazine to form a hydrazone. Hydroxide ion and heat differentiate the Wolff-Kishner Reduction from ordinary hydrazone formation. After the hydrazone is formed, OH– removes a proton from the NH2 group. Heat is required because these protons are not easily removed. Resonance places some of the negative charge on carbon, which abstracts a proton from water. The last two steps are repeated to from the deoxygenated product and N2 gas
The mechanism for Wolff-Kishner
List of Name Reaction of Organic Chemistry consist of detail Reaction Mechanism of all name reactions of Organic Chemistry.
Recent Concepts
- Aldol condensation
- Arndt−Ester synthesis
- Baeyer−Villiger Oxidation
- Benzoin Condensation
- Beckmann Rearrangement
- Cannizzaro Reaction
- Clemmensen Reduction
- Claisen condensation
- Etard’s Reaction
- Friedel-Crafts alkylation
- Friedel Crafts Acylation
- Fries Rearrangement
- Gattermann-Koch Reaction
- Grignard Reagent
- Hell-Volhard-Zelinsky Reaction
- Hunsdieker reaction
- Hoffmann Bromamide Degradation
- Jones reagent
- Kolbes Reaction
- Knoevenagel Reaction









