Kolbes Reaction
About Kolbes Reaction
Sodium phenoxide is heated with carbon dioxide at 120-140°C under pressure to yield sodium salicylate, which upon acidification produces salicylic acid is know as Kolbes reactions
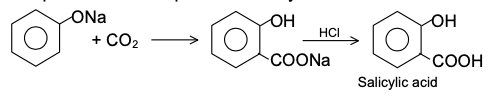
A small amount of p− derivative is also formed. If the temperature rises above 140° C, the p− isomer is the main product.
Mechanism of Kolbe's Reaction
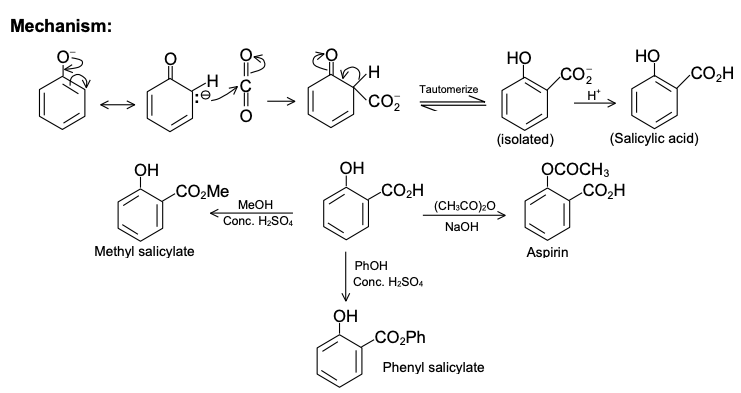
Salicylic acid is used for the preparation of Aspirin, oil of wintergreen (methyl salicylate), and salol (phenyl salicylate).
List of Name Reaction of Organic Chemistry consist of detail Reaction Mechanism of all name reactions of Organic Chemistry.
Recent Concepts
- Aldol condensation
- Arndt−Ester synthesis
- Baeyer−Villiger Oxidation
- Benzoin Condensation
- Beckmann Rearrangement
- Cannizzaro Reaction
- Clemmensen Reduction
- Claisen condensation
- Etard’s Reaction
- Friedel-Crafts alkylation
- Friedel Crafts Acylation
- Fries Rearrangement
- Gattermann-Koch Reaction
- Grignard Reagent
- Hell-Volhard-Zelinsky Reaction
- Hunsdieker reaction
- Hoffmann Bromamide Degradation
- Jones reagent
- Kolbes Reaction
- Knoevenagel Reaction









