Friedel Crafts Acylation
About Friedel-Crafts Acylation
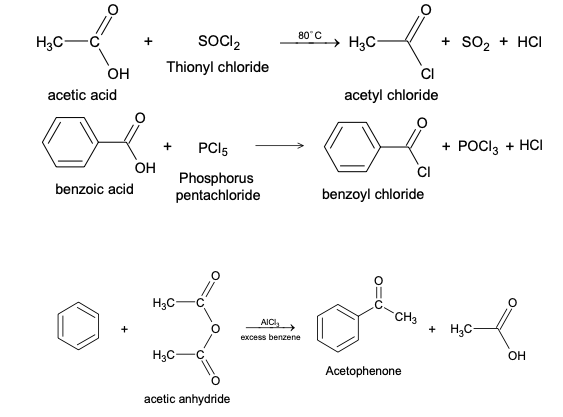
Substitution of hydrogen by acyl group in aromatic compound is known as Friedel-Crafts Acylation. The most commonly used catalyst is anhydrous AlCl3, although other Lewis acids can also be used. The acylating reagents are either acid halides or acid anhydrides.The Mechanism of Friedel-Crafts acylation involves following steps.The generation of attacking species may follow two different mechanisms.
Reaction Mechanism of Friedel-Crafts Acylation
The attacking species may also be 1 : 1 complex of AlCl3 with the acid halide. With acid anhydrides, free acyl cation is the attacking reagent generated by the attack of AlCl3.The substitution mechanism may be written as In either case the catalyst is required in considerable extent as the catalyst also forms complex with the ketone produced as a result of Lewis acid-base reaction.

The product ketone is then extracted with ice-cold dilute acidic solution.In presence of activating groups, the reaction takes place easily with good yield, whereas, deactivating groups inhibit the reaction. Hence, nitrobenzene is the commonly used solvent.
Reduction of Acyl to Alkene
When cyclic anhydrides are used as one of the component, Friedel-Crafts acylation provides a means of adding a new ring to an aromatic compound.
Factors Determining mechanism of alkylation :
(i) Nature of alkyl groups
If the alkyl group is simple CH3- or CH3CH2-, then a complex between alkyl halide and Lewis acid is the electrophile as shown in second mechanism. But because of the relative stability of s-and t- carbonium ions, the adducts with s- and t- alkyl halides ionise and it is now the s- and t- carbonium ion that is predominantly the active species. e.g.
Me3C-Cl + AlCl3 ---> . Me3C+ + . AlCl4
(ii) Nature of Lewis acid as catalyst
The order of effectiveness of Lewis acid catalysts has been shown to be
AlCl3 > FeCl3 > BF3 > TiCl3 > ZnCl2 > SnCl4
The action of Me3CCH2Cl/AlCl3 on benzene is found to yield almost completely the rearranged product, PhCMe2CH2Me, which can be explained on the basis of the initial electrophilic complex being polarized enough to allow the rearrangement of [Me3CCH2]d+-----Cl----AlCl3d- to the more stable [Me2CCH2Me]d+ -----Cl-----AlCl3d-. By contrast Me3CCH2Cl/FeCl3 on benzene is found to yield almost completely the unrearranged product, Me3CCH2Ph. This is due to the fact that the complex with the weaker Lewis acid, FeCl3, is not polarized enough to allow the rearrangement.
(iii) Temperature
Not only nature of the alkyl group, but also temperature determines the nature of electrophile. e.g. n-alkyl group can be introduced to a fair extent without rearrangement at low temperatures, because ionisation of the adduct is retarded. But at higher temperatures, carbonium ion is formed which rearranges and the product is rearranged alkyl benzene. Thus n-propylchloride gives isopropyl benzene.
Limitations of Friedel−Crafts Acylation
(i)Acylation requires more catalyst as the product ketone forms complex with the catalyst.
(ii)Acylation in presence of strong deactivating groups (like -NO2, -COCl, -SO3H, -CCl3 etc.) is not recommended as it gives very poor yield.
(iii)Acylation is not recommended in presence of -OH, -NH2, -NHR and -NR2 groups as they give O-acylated and N-acylated products.
The Friedel - Crafts acylation reaction is an effective means of introducing an acyl group into an aromatic ring. The reaction is often carried out by treating the aromatic compound with an acyl halide. Unless the aromatic compound is one that is highly reactive, the reaction requires the addition of at least one equivalent of a Lewis acid (such as AlCl3) as well. The product of the reaction is an aryl ketone. Moreover these groups also forms complexes with the catalyst thereby inhibiting the reaction.
Questions based on Friedel−Crafts Acylation
List of Name Reaction of Organic Chemistry consist of detail Reaction Mechanism of all name reactions of Organic Chemistry.
Recent Concepts
- Aldol condensation
- Arndt−Ester synthesis
- Baeyer−Villiger Oxidation
- Benzoin Condensation
- Beckmann Rearrangement
- Cannizzaro Reaction
- Clemmensen Reduction
- Claisen condensation
- Etard’s Reaction
- Friedel-Crafts alkylation
- Friedel Crafts Acylation
- Fries Rearrangement
- Gattermann-Koch Reaction
- Grignard Reagent
- Hell-Volhard-Zelinsky Reaction
- Hunsdieker reaction
- Hoffmann Bromamide Degradation
- Jones reagent
- Kolbes Reaction
- Knoevenagel Reaction









