Wurtz Reaction
About Wurtz Reaction
An ethereal solution of an alkyl halide (preferably the bromide or iodide) is treated with sodium, when alkane is obtained. For example,
.png)
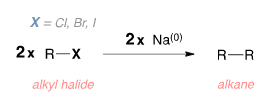
In this reaction, two R groups are coupled by reacting RBr, RCl or RI with Na or K. The yields of the product are best for 1° alkyl halide (60%) and least for 3° alkyl halides (10%).
Looking further in the above reaction, it was found that in addition to the desired alkane R1-R2, there will also be present the alkanes R1-R1 and R2-R2. Unsaturated hydrocarbons are also obtained. Obviously, then, the best yield of an alkane will be obtained when R1 & R2 are same, i.e., when the alkane contains an even number of carbon atoms and is symmetrical. It has been found that the Wurtz reaction gives good yields only for 'even carbon' alkanes of high molecular weight, and that the reaction generally fails with tertiary alkyl halides.
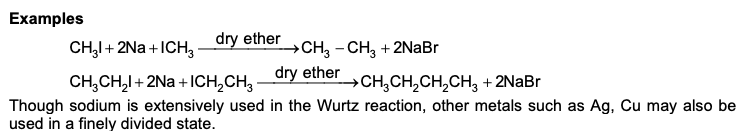
[Note: Metal other than sodium, which can be employed in Wurtz reaction are Ag and Cu in finely divided state]
Reaction Mechanism of Wurtz Reaction
The reaction involves the intermediate formation of free radicals. The suggested mechanism for the Wurtz reaction is shown as

Example of Wurtz Reaction
Limitation of Wurtz Reaction
Application of Wurtz Reaction
Wurtz reaction is used to ascend the homologous series through the preparation of higher alkanes containing even number of carbon atoms.
List of Name Reaction of Organic Chemistry consist of detail Reaction Mechanism of all name reactions of Organic Chemistry.
Recent Concepts
- Aldol condensation
- Arndt−Ester synthesis
- Baeyer−Villiger Oxidation
- Benzoin Condensation
- Beckmann Rearrangement
- Cannizzaro Reaction
- Clemmensen Reduction
- Claisen condensation
- Etard’s Reaction
- Friedel-Crafts alkylation
- Friedel Crafts Acylation
- Fries Rearrangement
- Gattermann-Koch Reaction
- Grignard Reagent
- Hell-Volhard-Zelinsky Reaction
- Hunsdieker reaction
- Hoffmann Bromamide Degradation
- Jones reagent
- Kolbes Reaction
- Knoevenagel Reaction









