
Naming Of Bigger Compounds
GOC of Class 11
Naming Of Bigger Compounds
Having learnt the names of simple compounds we are now in happy situation to proceed to name the bigger or complex compounds by IUPAC system. There are however more than 70 rules in the system which guide us to name the organic compounds but here we are required to know only six basic rules.
- Rule−1: Select the longest continuous carbon chain in a molecule whether it is in straight line (I) or not in straight line (II).
-
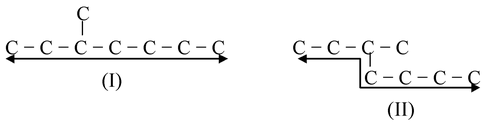
- Rule−2: If the compounds contains one or more longest continuous carbon chains then that chain is accepted which has the largest number of branches.
- Let us understand it through an example,
-
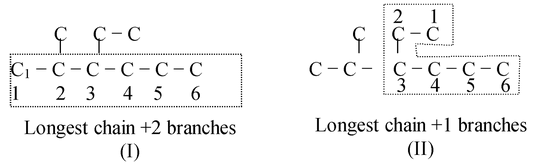
- The above two structural formulae are identical with, difference that if we view it in two ways (I) and (II). Both have the longest chains of 6 carbons atoms, which are numbered. In viewing as (I) we get two branches while viewed as (II) we get one branch.
- So, (I) is the correct longest chain.
- Rule−3: The rule states that after selecting the longest carbon chain, lowest position number is to be given to the position of substituent say, S. Let us have an example
-
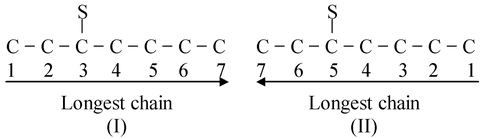
- In (I) 3 is the lower. Hence (I) is accepted and (II) is rejected. The positional number is also termed as LOCANT which means a number that locates the position of a substituent eg. Locant of S in (I)=3 and locant of S in (II)=5 due to incorrect numbering.
- Rule−4: This rule is applicable to those compounds which have more than one substituent(S). In such cases counting is done from that terminal of carbon chain which gives lesser value for the sum of locant values of substituents. Such as
-
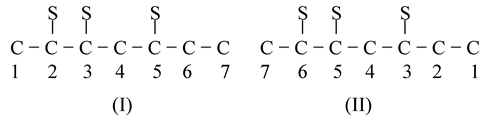
- 2 + 3 + 5 = 10 3 + 5 + 6 = 14
- The compounds are identical and after counting we get 7C−atoms. But after counting, the sum of the locants of S should be lowest. Hence (I) is accepted for naming the compound.
<
IUPAC names of compounds containing poly functional groups
| Functional groups | Formula | Name as substituent | Name as parent |
| Acid | −COOH | Carboxy |
Carboxylic or −oic
acid |
| Sulphonic acid | −SO3H | Sulpho | Sulphonic acid |
| Ester |
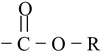 |
Alkoxycarbonyl | –oate |
| Anhydride |
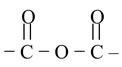 |
– | –oic anhydride |
| Acid halide | −COX | Halo carbonyl | –oyl halide |
| Acid amide | −CONH2 | Carbamoyl or Amido | Amide |
| Cyanide | −C ≡ N | Cyano | Nitrile |
| Iso cyanide |
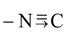 |
Iso cyano |
Iso nitrile or carbyl
amine |
| Aldehyde | −CHO |
Oxo, aldo (for aldehyde)
Or formyl (for CHO) |
–al |
| Ketone |
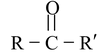 |
Oxo or keto | –one |
| Alcohol | −OH | Hydroxy | –ol |
| Thiol | –SH | Mercapto | Thiol |
| Amines | −NH2 | Amino | Amine |
| Ethers | −O− | Alkoxy | − |
| Alkene | = | – | ene |
| Alkyne | ≡ | – | yne |
| Nitro. | –NO2 | Nitro | – |
| Nitroso. | –N = O | Nitroso | – |
| Halogen | −X | Halo | − |
| Alkyl | –R | Alkyl | – |
Talk to a counsellorHave doubts? Our support team will be happy to assist you!

Free Learning Resources
PW Books
Notes (Class 10-12)
PW Study Materials
Notes (Class 6-9)
Ncert Solutions
Govt Exams
Class 6th to 12th Online Courses
Govt Job Exams Courses
UPSC Coaching
Defence Exam Coaching
Gate Exam Coaching
Other Exams
Know about Physics Wallah
Physics Wallah is an Indian edtech platform that provides accessible & comprehensive learning experiences to students from Class 6th to postgraduate level. We also provide extensive NCERT solutions, sample paper, NEET, JEE Mains, BITSAT previous year papers & more such resources to students. Physics Wallah also caters to over 3.5 million registered students and over 78 lakh+ Youtube subscribers with 4.8 rating on its app.
We Stand Out because
We provide students with intensive courses with India’s qualified & experienced faculties & mentors. PW strives to make the learning experience comprehensive and accessible for students of all sections of society. We believe in empowering every single student who couldn't dream of a good career in engineering and medical field earlier.
Our Key Focus Areas
Physics Wallah's main focus is to make the learning experience as economical as possible for all students. With our affordable courses like Lakshya, Udaan and Arjuna and many others, we have been able to provide a platform for lakhs of aspirants. From providing Chemistry, Maths, Physics formula to giving e-books of eminent authors like RD Sharma, RS Aggarwal and Lakhmir Singh, PW focuses on every single student's need for preparation.
What Makes Us Different
Physics Wallah strives to develop a comprehensive pedagogical structure for students, where they get a state-of-the-art learning experience with study material and resources. Apart from catering students preparing for JEE Mains and NEET, PW also provides study material for each state board like Uttar Pradesh, Bihar, and others
Copyright © 2026 Physicswallah Limited All rights reserved.