
Resonance Effect Of Mesomerism
GOC of Class 11
Resonance Effect Of Mesomerism
There are many organic molecules which can not be represented by a single lewis structure. In turn, they are assigned more than one structure called canonical forms or contributing or resonating structures. The phenomenon exhibited by such compounds is called resonance. For example, 1, 3-Butadiene has following resonance structures.
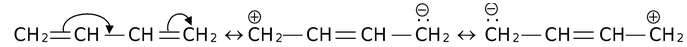
and canonical forms of vinyl chloride are
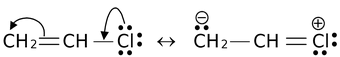
While drawing these canonical forms, the prime thing that has to be kept in mind is that the relative position of any of the atom should not change while we are allowed to change the relative positions of π-bond electron pairs or lone pairs or distribution of charge to other atoms. Also remember that it is not the case that some molecules have one canonical form and some have another form. All the molecules of the substance have the same structure. That structure is always the same all the time and is a weighted average of all the canonical forms. In real sense, these canonical forms have no existence except in our imaginations.
Now we are in a position to discuss about the conditions necessary for a compound to show resonance. The two essential conditions are
(a) There must be conjugation in the molecule. The conjugation is defined as the presence of alternate double and single bonds in the compound like 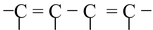 .
.
(b )The part of the molecule having conjugation must be essentially planar or nearly planar.
The first condition of conjugation is not only confined to the one mentioned above but some other systems are also categorized under conjugation. These are
(i )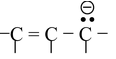
(ii) 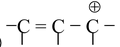
(iii) 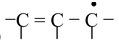
(iv) 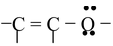
(v) 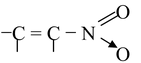
So, any molecule satisfying both the conditions will show resonance. For example, we consider phenol. The structure of phenol is
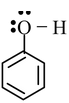
By looking at the structure, it must be clear to you that the compound possess conjugation of the type  as well as the category (iv) because the lone pairs on oxygen are in conjugation with unsaturated
as well as the category (iv) because the lone pairs on oxygen are in conjugation with unsaturated
(sp2 hybridised) carbon of the ring. Since, oxygen atom is sp3 hybridised in phenol, the lone pairs on oxygen are nearly planar with respect to the Pz orbital of carbon linked to oxygen. Thus, both the conditions are fulfilled by phenol, therefore it does show resonance and its resonance structures are represented as
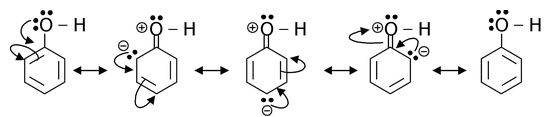
This has to be borne in mind that resonance always results in different distribution of electron density than would be the case if there were no resonance. It is a permanent effect, also referred as mesomeric effect. Resonance (mesomeric) effect is of two types
(i)If the atom or group of atom is giving electrons through resonance, it is called +R or +M effect. For example,
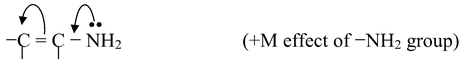
Other groups that shows +M effect are −NHR, −NR2, −OH, −OR, −NHCOR, −Cl, −Br, −I etc.
(ii)If the atom or group of atom is withdrawing electrons through resonance, it is called −R or −M effect. For example,
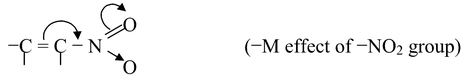
Other groups showing −M effect are −CN, −CHO, −COR, −CO2H, −CO2R, −CONH2, −SO3H, −COCl etc.
Now, let us consider resonance in nitrobenzene and its various canonical structures are
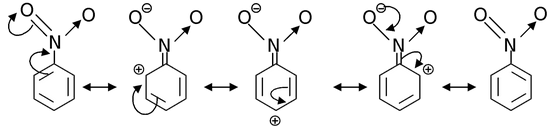
The –NO2 group in nitrobenzene has –M effect. In general, if any atom (of the group) attached to the carbon of benzene ring bears atleast one lone pair, then the group shows +M effect while if the atom (of the group) linked to the benzene carbon bears either a partial or full positive charge, then the group exhibits −M effect.
In drawing the canonical forms and deciding about their relative stabilities, following rules are given for your guidance.
(i) All the canonical forms must be bonafide lewis structures. For example, none of them may have a carbon with five bonds
(ii) All atoms taking part in the resonance must lie in a plane or nearly so. The reason for planarity is to have maximum overlap of the p-orbitals.
(iii) All canonical forms must have the same number of unpaired electrons. Thus
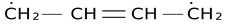 is not a valid canonical form for 1,3−butadiene.
is not a valid canonical form for 1,3−butadiene.
(iv) The energy of the hybrid (actual) molecule is lower than that of any canonical form. Obviously then, delocalization is a stabilizing phenomenon. The difference in energy between the hybrid and the most stable canonical structure is called resonance energy.
(v) All canonical forms do not contribute equally to the actual molecule. Each form contributes in proportion to its stability, the most stable form contributing the most.
(vi) Structures with more covalent bonds are generally more stable than those with fewer covalent bonds.
(vii) Structures with formal charges are less stable than uncharged structures. For charged structures, the stability is decreased by an increase in charge separation and the structure with two like charges on adjacent atoms are highly unfavourable.
(viii) Structures that carry a negative charge on a more electronegative atom are more stable than those in which the charge is on a less electronegative atom. For example,
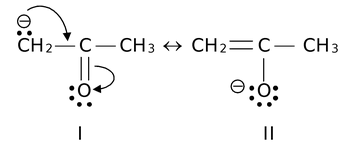
Structure II is more stable than I. Similarly positive charges are best occupied on atoms of low electronegativity.
(ix) Those structures in which octet of every atom (except for hydrogen which have duplet) is complete are more stable than the others with non-complete octets. For example,
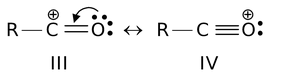
Structure IV is more stable than III.
